
Concept explainers
(a)
Interpretation:
It is to be determined if the given pair is not resonance structures of one another.
Concept introduction:
Resonance exists in species for which there are two or more valid Lewis structures. Resonance structures differ only in the placement of their electrons, not their atoms. Resonance stabilization is usually high when the resonance contributors are equivalent. More the number of the resonance contributors, the more is the resonance stabilization.
Answer to Problem 1.75P
The given structures are not resonance structures of one another.
Explanation of Solution
The structures of the given pair are shown below:
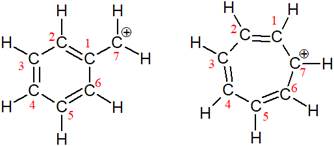
The two structures have completely different carbon skeleton. The first one has a six membered ring while the second has a seven membered ring. Since these two structures do not have the same position of the atoms, they cannot be resonance structures.
From the position of atoms and electrons, it is found that the given pair is not resonance structures of one another.
(b)
Interpretation:
It is to be determined if the given pair is not resonance structures of one another.
Concept introduction:
Resonance exists in species for which there are two or more valid Lewis structures. Resonance structures differ only in the placement of their electrons, not their atoms. Resonance stabilization is usually high when the resonance contributors are equivalent. More the number of the resonance contributors, the more is the resonance stabilization.
Answer to Problem 1.75P
The given structure is not resonance structures of one another.
Explanation of Solution
The structures of the given pair are shown below:
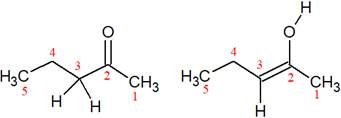
In these two structures, the position of the hydrogen atoms is not the same. Hence, these structures cannot be resonance structures of each other.
From the position of atoms and valence electrons, it is found that the given pair is not resonance structures of one another.
(c)
Interpretation:
It is to be determined if the given pair is not resonance structures of one another.
Concept introduction:
Resonance exists in species for which there are two or more valid Lewis structures. Resonance structures differ only in the placement of their electrons, not their atoms. Resonance stabilization is usually high when the resonance contributors are equivalent. More the number of the resonance contributors, the more is the resonance stabilization.
Answer to Problem 1.75P
The given structure is resonance structures of one another.
Explanation of Solution
The structures of the given pair are as shown below.
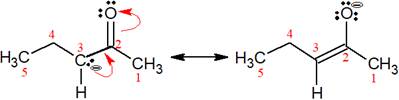
To obtain the second resonance structure, two curved arrows are drawn and the electrons are moved accordingly. The two structures differ only in the placement of their electrons, not their atoms. Therefore, the given pair is resonance structures of one another.
From the position of atoms and valence electrons, it is found that the given pair is resonance structures of one another.
(d)
Interpretation:
It is to be determined if the given pair is not resonance structures of one another.
Concept introduction:
Resonance exists in species for which there are two or more valid Lewis structures. Resonance structures differ only in the placement of their electrons, not their atoms. Resonance stabilization is usually high when the resonance contributors are equivalent. More the number of the resonance contributors, the more is the resonance stabilization.
Answer to Problem 1.75P
The given structure is resonance structures of one another.
Explanation of Solution
The structures of the given pair are shown below:
To obtain the second resonance structure, two curved arrows are drawn and the valence electrons are moved accordingly. Similarly, to arrive at the third resonance structure, two curved arrows are drawn and valence electrons are moved accordingly. The resonance structures differ only in the placement of their electrons, not their atoms. Therefore, the given pair is resonance structures of one another.
From the position of atoms and valence electrons, it is found that the given pair is resonance structures of one another.
(e)
Interpretation:
It is to be determined if the given pair is not resonance structures of one another.
Concept introduction:
Resonance exists in species for which there are two or more valid Lewis structures. Resonance structures differ only in the placement of their electrons, not their atoms. Resonance stabilization is usually high when the resonance contributors are equivalent. More the number of the resonance contributors, the more is the resonance stabilization.
Answer to Problem 1.75P
The given structure is resonance structures of one another.
Explanation of Solution
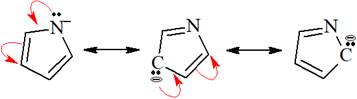
The structures of the given pair are shown below:
![]()
To obtain the second resonance structure, a curved arrow is drawn and the valence electrons are moved accordingly. Similarly, to arrive at the third resonance structure, two curved arrows are drawn and valence electrons are moved accordingly. The resonance structures differ only in the placement of their electrons, not their atoms. Therefore, the given pair is resonance structures of one another.
From the position of atoms and valence electrons, it is found that the given pair is resonance structures of one another.
(f)
Interpretation:
It is to be determined if the given pair is not resonance structures of one another.
Concept introduction:
Resonance exists in species for which there are two or more valid Lewis structures. Resonance structures differ only in the placement of their electrons, not their atoms. Resonance stabilization is usually high when the resonance contributors are equivalent. More the number of the resonance contributors, the more is the resonance stabilization.
Answer to Problem 1.75P
The given structure is not resonance structures of one another.
Explanation of Solution
The structure of the given pair is as follows:
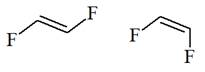
Resonance structures differ only in the placement of their electrons, not their atoms. In the given pair, the placement of electrons is the same. Therefore, the given pair is not resonance structures of one another.
From the position of atoms and valence electrons, it is found that the given pair is not resonance structures of one another.
(g)
Interpretation:
It is to be determined if the given pair is not resonance structures of one another.
Concept introduction:
Resonance exists in species for which there are two or more valid Lewis structures. Resonance structures differ only in the placement of their electrons, not their atoms. Resonance stabilization is usually high when the resonance contributors are equivalent. More the number of the resonance contributors, the more is the resonance stabilization.
Answer to Problem 1.75P
The given structure is not resonance structures of one another.
Explanation of Solution
The structure of the given pair is as follows:
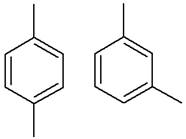
Resonance structures differ only in the placement of their electrons, not their atoms. In the given pair, the placement of electrons is the same. Therefore, the given pair is not resonance structures of one another.
From the position of atoms and valence electrons, it is found that the given pair is resonance structures of one another.
(h)
Interpretation:
It is to be determined if the given pair is not resonance structures of one another.
Concept introduction:
Resonance exists in species for which there are two or more valid Lewis structures. Resonance structures differ only in the placement of their electrons, not their atoms. Resonance stabilization is usually high when the resonance contributors are equivalent. More the number of the resonance contributors, the more is the resonance stabilization.
Answer to Problem 1.75P
The given structure is not resonance structures of one another.
Explanation of Solution
The structure of the given pair is as follows:
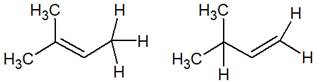
Resonance structures differ only in the placement of their electrons, not their atoms. In the given pair, the placement of electrons is different; at the same time, one H atom changes its position. Therefore, the given pair is not resonance structures of one another.
From the position of atoms and valence electrons, it is found that the given pair is not resonance structures of one another.
Want to see more full solutions like this?
Chapter 1 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Draw two resonance structures of the given molecule/ion. Be sure to include all lone pairs. :Ö:arrow_forwardDraw a Lewis structure for thiocyanic acid, HSCN, adding charges and lone electron pairs to the appropriate atoms. Explicitly draw all H atoms. Include all valence lone pairs in your answer. opy astearrow_forwardA resonance hybrid is a structure that can be depicted by more than one valid Lewis structure. part1: Draw the major resonance form of fulminic acid, HCNO, with the atoms connected as indicated in the formula. Your structure should have nonzero formal charges minimized, and it should include all nonzero formal charges and all nonbonding electrons. part2: Draw the second most important resonance form of fulminic acid, HCNO, with the atoms connected as indicated in the formula. Your structure should have nonzero formal charges minimized, and it should include all nonzero formal charges and all nonbonding electrons.arrow_forward
- A resonance hybrid is a structure that can be depicted by more than one valid Lewis structure. part1: Draw the major resonance form of fulminic acid, HCNO, with the atoms connected as indicated in the formula. Your structure should have nonzero formal charges minimized, and it should include all nonzero formal charges and all nonbonding electrons. part2: Draw the second most important resonance form of fulminic acid, HCNO, with the atoms connected as indicated in the formula. Your structure should have nonzero formal charges minimized, and it should include all nonzero formal charges and all nonbonding electrons. part3: Draw the least important resonance contributor for fulminic acid, HCNO, with the atoms connected as indicated in the formula. Your structure should have nonzero formal charges minimized and should include all nonzero formal charges and all nonbonding electrons.arrow_forwardFollow the curved arrows shown in the image below and modify to draw the correct second resonance structure. 4 DE C C Xarrow_forwardDraw a Lewis structure for cyanide ion, CN¯, adding charges and lone electron pairs to the appropriate atoms. Explicitly draw all H atoms. Include all valence lone pairs in your answer. C opy aste :c=Narrow_forward
- Which of the following resonance structure for OCN- will contribute most to the correct structure of OCN-? O = C-N, with one lone pair on O and three lone pairs on N O=C=N, with two lone pairs on O, and two lone pairs on N O=C=N, with one lone pair on O, two lone pairs on C, and one lone pair on N O-C = N, with three lone pairs on O and one lone pair on N They All contribute equally to the structure on OCN-arrow_forwardWhich of the following species is a valid resonance structure of A? Usecurved arrows to show how A is converted to any valid resonancestructure. When a compound is not a valid resonance structure of A,explain why not.arrow_forwardComplete the following Lewis structure by adding in missing lone pairs and pi bonds. Assume all atoms are neutral (having no formal charge). Do not add additional atoms to the structure. H, F H' Br H-C- I + I : 0arrow_forward
- Consider the resonance structures of the ion shown below. In the actual overall resonance hybrid, which carbon atom will have the greatest electropositive character (i.e. the largest 8+)? Select an answer and submit. For keyboard navigation, use the up/down arrow keys to select an answer. a C1 b C2 C d e C3 C4 C5 f C6 6 (+) 5 2 4 3 -CIarrow_forwardAdd the number of missing lone pairs for the assigned atoms (H is not included) HC NO 4 0 3 1 5 NO 0 CH3arrow_forwardNhy is resonance structure A and resonance structure B not the same structure? If you rotate the molecule like a spicket handle (l. e. clockwise with the C at the center and the R group remaining in place as you look down the C - R bond) wouldn't you get the same thing? Why are these not equivalent structures? Resonance structure A t Resonance structure B Actual structurearrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

