
Repeat Prob. 8–114 if heat were supplied to the pressure cooker from a heat source at 180°C instead of the electrical heating unit?
8–114 A 4-L pressure cooker has an operating pressure of 175 kPa. Initially, one-half of the volume is filled with liquid water and the other half by water vapor. The cooker is now placed on top of a 750-W electrical heating unit that is kept on for 20 min. Assuming the surroundings to be at 25°C and 100 kPa, determine (a) the amount of water that remained in the cooker and (b) the exergy destruction associated with the entire process.
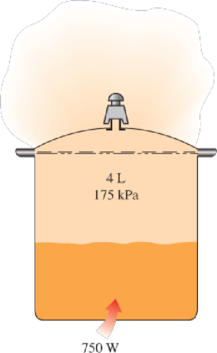
FIGURE P8–114
(a)
The final mass of water that remained in the cooker.
Answer to Problem 115RP
The final mass of water that remained in the cooker is
Explanation of Solution
Express the mass balance for a pressure cooker which acts as a system.
Here, initial mass is
Write an energy balance for a system.
Here, internal energy at state 1 and 2 is
Calculate the initial mass in the tank
Here, saturated liquid specific volume is
Calculate the initial internal energy
Here, specific internal energy of saturated fluid is
Calculate the initial entropy
Here, specific entropy of saturated fluid is
Write the internal energy at state 2
Here, dryness fraction at state 2 is
Write the specific volume at state 2
Write the formula to calculate the mass of the water remained in tank
Here, volume of the cooker is
Conclusion:
From the Table A-5 of “Saturated water: Pressure”, obtain the saturated liquid specific volume
From the Table A-5 of “Saturated water: Pressure”, obtain the enthalpy
Since one half of the volume is filled with liquid water and other half by water vapor, calculate the volume of tank
Substitute
Substitute 1.893 kg for
Substitute 1.893 kg for
Substitute
Substitute
Re-write the Equation (II) using the Equation (I),
Substitute
Substitute
Substitute 0.001918 for
Substitute
Thus, the final mass of water that remained in the cooker is
(b)
The amount of exergy destructed during the heat supplied to pressure cooker from heat source.
Answer to Problem 115RP
The amount of exergy destructed during the heat supplied to pressure cooker from heat source is
Explanation of Solution
For an extended system, write the simplification rate form of the entropy balance.
Here, entropy generation is
Calculate the exergy destroyed during the process
Here, dead state temperature is
Substitute Equation (XI) in Equation (XII).
Calculate the entropy at state 2
Conclusion:
Substitute 0.001918 for
Substitute 1.8945 kg for
Substitute 298 K for
Thus, the amount of exergy destructed during the heat supplied to pressure cooker from heat source is
Want to see more full solutions like this?
Chapter 8 Solutions
Thermodynamics: An Engineering Approach
- Refrigerant R-134a enters the compressor of a refrigeration machine at 140 kPa pressure and -10°C temperature and exits at 1 MPa pressure. The volumetric flow of the refrigerant entering the compressor is 0.23 m3/minute. The refrigerant enters the throttling valve at 0.95 MPa pressure and 30°C, exiting the evaporator as saturated steam at -18°C. The adiabatic efficiency of the compressor is 78%. Show the cycle in the T-s diagram. a) Calculate the power required to start the compressor. b) Calculate the heat taken in a unit time from the cooled environment. COP=? c) Calculate between the evaporator and the compressor, how much the pressure of the refrigerant drops, and how much is the heat gain.arrow_forwardA schematic of a steam power plant is shown in the figure. The boiler volume is 50 liter and contains 90% liquid and 10% steam at 100kPa. The boiler is heated and when the pressure at the boiler reaches 700kPa, the pressure valve is switched on to maintain a constant pressure of 700kPa. At this pressure, saturated steam moves to an adiabatic turbine. The saturated steam leaves the turbine at 100kPa to the atmosphere. When all the liquid at the boiler evaporator, the heating stops automatically. Calculate the amount of work obtained by the turbine and the amount of heat to be provided to the boiler over one cycle.arrow_forwardAn air conditioner with refrigerant-134a as the refrigerant is used to keep a large space at 20°C by rejecting the waste heat to the outside air at 37 °C. The room is gaining heat through the walls and the windows at a rate of 125 kJ/min while the heat generated by the computer, TV, and lights amounts to 0.7 kW. Unknown amount of heat is also generated by the people in the room. The condenser and evaporator pressures are 1200 and 500 kPa, respectively. The refrigerant is saturated liguid at the condenser exit and saturated vapor at the compressor inlet. If the refrigerant enters the compressor at a rate of 65 L/min and the isentropic efficiency of the compressor is 70%, determine (a) the temperature of the refrigerant at the compressor exit, (b) the rate of heat generated by the people in the room, (c) the COP of the air conditioner, and (d) the minimum volume flow rate of the refrigerant at the compressor inlet for the same compressor inlet and exit conditions.arrow_forward
- A boiler produces 5.4 tonnes/hour of steam at a pressure of 1.5 MPa and a temperature of 350°C. Feedwater enters at a temperature of 44.5°C. At exit from the economizer part of the boiler the temperature is 93.5°C. At exit from the evaporator part of the boiler the steam is 90 A % dry. Energy is supplied by 600 kg of coal per hour, which has a calorific value of 32 MJ/kg. The A/F ratio is 15 1. The temperature of the flue gas at exit from the the economizer part of the boiler is 210°C. The average specific heat at constant pressure of the in the economizer is 1045 J/kg.K. When the ambient temperature is 24°C : 7. IS flue gas 7.1 Calculate the efficiency of the boiler. [83.3 %] Calculate the equivalent evaporation from and at 100°C per unit mass of fuel. |11.9| Draw up an energy balance, on a kJ/kg coal basis, with percentages of the total energy supplied. Jeconomizer 6.8 %, evaporator 62 %, superheater 15.5 %, to atmosphere 9.7 %, other 7.2 7.3 losses 7 %]arrow_forwardb) A desktop computer is to be cooled by a fan. The electronic components of the computer consume 100 W of power under full-load conditions. The computer is to operate in environments at temperatures up to 55°C and at elevations up to 3000 m where the atmospheric pressure is 70.12 kPa. The exit temperature of air is not to exceed 65°C to meet the reliability requirements. Also, the average velocity of air is not to exceed 110 m/min at the exit of the computer case, where the fan is installed to keep the noise level down. Calculate the required flow rate and the diameter of the casing of the fan that needs to be installed.arrow_forward1. A Carnot heat engine receive 1200 kJ of heat per cycle from a high-temperature reservoir at 420 °C and reject heat to a low-temperature reservoir at 35 °C. Determine the thermal efficiency of this Carnot engine, the maximum work produced and the amount of heat rejected to the low temperature reservoir. II. A 1-m³ tank containing air at 25°C and 500 Kpa (abs) is connected through a valve to another tank containing 5 kg of air at 35°C and 200 Kpa (abs). Now the valve is opened, and the system is allowed to reach thermal equilibrium, which are at 20 °C. Determine the volume of the second tank and the final equilibrium pressure of air.arrow_forward
- An air refrigeration system having pressure ratio of 5 takes air at 0°C and 1 atm. It is compressed and then cooled to 19°C. A pressure drop of 5 kPa is measured in both cooler and refrigerator. If the efficiency of the compressor is 95% and that of the expander is 75%, determine the refrigeration capacity of the system TR, and the COP of the system if the flow of air is 7.5 kg/min. Assume compression and expansion processes to be isentropic.arrow_forwardSolve the following problems: Note: Draw the p-h diagram completely labeling the respective refrigerant conditions (pressures and temperatures) and define the properties of refrigerant (enthalpies and specific volume) needed to solve the respective problems. In an ammonia refrigeration system, the capacity is (1) 210 kW at a temperature of-20°C. The vapor from the evaporator is pumped by one compressor to the condensing pressure of 1431 kPa. Later, the system was revised to a two-stage compression operating on the cycle shown below with intercooling but no removal of flash at 555 kPa. (а) Calculate the power required by the single compressor in the original system. (b) Calculate the total power required by the two compressors in the revised system. Condenser 1431kPa High-stage compressor Intercooler 555kPa Evaporator 2í0kW -20°C Low-stage compresaorarrow_forwardTHERMOFLUID In steam power plants, open feedwater heaters are frequently utilized to heat the feedwater by mixing it with steam bled off the turbine at some intermediate stage. Consider an open feedwater heater that operates at a pressure of 1000 kPa. Feedwater at 50°C and 1000 kPa is to be heated with superheated steam at 200°C and 1000 kPa. In an ideal feedwater heater, the mixture leaves the heater as saturated liquid at the feedwater pressure. Determine the ratio of the mass flow rates of the feedwater and the superheated vapor for this case.arrow_forward
- A refrigeration plant works between the temperatures limits of -5°C and 25°C. The refrigerant is CO2 is wet at the entry to the compressor and has dryness fraction of 0.6. The refrigerator has actual COP 70% of the theoretical COP. If there is no under-cooling, determine the ice formed during a period of 24 hours from 20°C. The mass of CO2 circulated is 5kg/min. Take enthalpy of fusion of ice as 336 kJ/kg. The properties of CO2 are as follows: Saturation temperature (C°) Specific enthalpy (kJ/kg) hf Specific entropy (kJ/kg-K) hg Sf 25 81.25 202.75 0.2513 -5 -7.53 238.5 -0.04187arrow_forwardA refrigeration plant works between the temperatures limits of -5°C and 25°C. The refrigerant is CO2 is wet at the entry to the compressor and has dryness fraction of 0.6. The refrigerator has actual COP 70% of the theoretical COP. If there is no under-cooling, determine the ice formed during a period of 24 hours from 20°C. The mass of CO2 circulated is 5kg/min. Take enthalpy of fusion of ice as 336 kJ/kg. The properties of CO2 are as follows:arrow_forwardIn steam power plants, open feedwater heaters are frequently utilized to heat the feedwater by mixing it with steam bled off the turbine at some intermediate stage. Consider an open feedwater heater that operates at a pressure of 1200 kPa. Feedwater at 50°C and 1200 kPa is to be heated with superheated steam at 200°C and 1200 kPa. In an ideal feedwater heater, the mixture leaves the heater as saturated liquid at the feedwater pressure. Determine the ratio of the mass flow rates of the feedwater and the superheated vapor for this case.arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





