
Concept explainers
a)
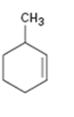
Interpretation:
The name of the cycloalkane shown is to be given.
Concept introduction:
The maximum number of carbons in the ring is counted. Based on the name of the parent cycloalkane–the cycloalkene is named with the suffix–ene. The cycloalkane is numbered such that the double bond is in between C1 & C2 and the first substituent has the lowest number possible. Usually the position of a double bond is not shown in the name because it is always between C1 & C2. In dienes and trienes, however the position of double bonds is shown.
To name:
The cycloalkane shown.
b)
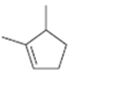
Interpretation:
The name of the cycloalkane shown is to be given.
Concept introduction:
The maximum number of carbons in the ring is counted. Based on the name of the parent cycloalkane–the cycloalkene is named with the suffix–ene. The cycloalkane is numbered such that the double bond is in between C1 & C2 and the first substituent has the lowest number possible. Usually the position of a double bond is not shown in the name because it is always between C1 & C2. In dienes and trienes, however the position of double bonds is shown.
To name:
The cycloalkane shown.
c)
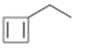
Interpretation:
The name of the cycloalkane shown to be given.
Concept introduction:
The maximum number of carbons in the ring is counted. Based on the name of the parent cycloalkane the cycloalkene is named with the suffix–ene. The cycloalkane is numbered such that the double bond is in between C1 & C2 and the first substituent has the lowest number possible. Usually the position of a double bond is not shown in the name because it is always between C1 & C2. In dienes and trienes, however the position of double bonds is shown.
To name:
The cycloalkane shown.
d)
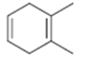
Interpretation:
The name the cycloalkane shown to be given.
Concept introduction:
The maximum number of carbons in the ring is counted. Based on the name of the parent cycloalkane–the cycloalkene is named with the suffix–ene. The cycloalkane is numbered such that the double bond is in between C1 & C2 and the first substituent has the lowest number possible. Usually the position of a double bond is not shown in the name because it is always between C1 & C2. In dienes and trienes, however the position of double bonds is shown.
To name:
The cycloalkane shown.
e)
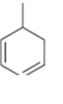
Interpretation:
The name the cycloalkane shown is to be given.
Concept introduction:
The maximum number of carbons in the ring is counted. Based on the name of the parent cycloalkane–the cycloalkene is named with the suffix–ene. The cycloalkane is numbered such that the double bond is in between C1 & C2 and the first substituent has the lowest number possible. Usually the position of a double bond is not shown in the name because it is always between C1 & C2. In dienes and trienes, however the position of double bonds is shown.
To name:
The cycloalkane shown
f)
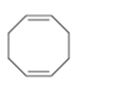
Interpretation:
The name of the cycloalkane shown is to be given.
Concept introduction:
The maximum number of carbons in the ring is counted. Based on the name of the parent cycloalkane–the cycloalkene is named with the suffix–ene. The cycloalkane is numbered such that the double bond is in between C1 & C2 and the first substituent has the lowest number possible. Usually the position of a double bond is not shown in the name because it is always between C1 & C2. In dienes and trienes, however the position of double bonds is shown.
To name:
The cycloalkane shown.
Trending nowThis is a popular solution!

Chapter 7 Solutions
Organic Chemistry
- How many alkene products are possible in the following reaction? CI CH3 NaOEt {"CH3 Harrow_forwardName the following cycloalkane with alkyl groups C2H5 C2H5 キ H CH3 C-H Н—с С —н CI- CH3 H CH3 CH3 CH3 CH3arrow_forwardThe following alkene is: O primary O monosubstituted O disubstituted secondaryarrow_forward
- Which one alkene will exhibit cis-trans isomerism? A B OC OA u B Darrow_forwardName the alkenes below. Use ONLY E/Z designators to indicate stereochemistry. H3C H Ja H CH₂-C H CH3 CH₂CH3arrow_forwardWhat is the IUPAC name of the following molecule? s O 1-cyclohexyl-1,3,4-trimethyl-(1Z,3E)-1,3-hexadiene 6-cyclohexyl-3,4,6-trimethyl-(3E,5Z)-3,5-hexadiene O 6-cyclohexyl-3,4,6-trimethyl-(3Z,5E)-3,5-hexadiene 1-cyclohexyl-1,3,4-trimethyl-(1E,3Z)-1,3-hexadienearrow_forward
- Which one of the following alkyl groups is the ethyl group? CH3 CH3- CH3CH,- CH3CH,CH,- CH3-C- CH3 CH3CHCH3 А B C E O A O E D.arrow_forwardWhat are the major and minor products formed when cyclohexene reacts with Br2 in the solvent CH2Cl2?arrow_forwardWhich of the following C6H12 alkenes is most stable?arrow_forward
- Draw the structures of the following molecules: (a) trans-l-Bromo-3-methylcyclohexane (b) cis-1, 2-Dimethylcyclobutane (c) trans-1 -tert-Butyl-2-ethylcyclohexanearrow_forwardName the alkene below. Use ONLY E/Z designators to indicate stereochemistry. H CH3 H3C C=C H2C=CHCHÇH H ČH3arrow_forwardName the alkene below. Use ONLY E/Z designators to indicate stereochemistry. H H H3C CH3 CH;CH2CH2 CH3arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

