
Concept explainers
a)
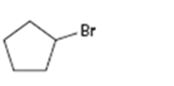
Interpretation:
The alkene that will give 1-bromocyclopentane in an addition reaction is to be identified.
Concept introduction:
In addition reactions small molecules like HCl, H2O, HBr etc. add to the double bond in an alkene to yield a single product. The addition to unsymmetrical
To state:
Which alkene will give 1-bromocyclopentane as product in an addition reaction.
b)
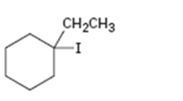
Interpretation:
The alkene that will give 1-ethyl-1-iodocyclopentane in an addition reaction is to be identified.
Concept introduction:
In addition reactions small molecules like HCl, H2O, HBr etc. add to the double bond in an alkene to yield a single product. The addition to unsymmetrical alkenes will follow Markovnikov’s rule which states that “in the addition of HX to an alkene, the H adds to the carbon with fewer alkyl substituents and X adds to the carbon with more alkyl substituents”.
To state:
Which alkene will give 1-ethyl-1-iodocyclohexane as product in an addition reaction.
c)
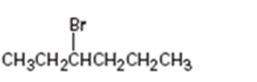
Interpretation:
The alkene that will give 3-bromohexane in an addition reaction is to be identified.
Concept introduction:
In addition reactions small molecules like HCl, H2O, HBr etc. add to the double bond in an alkene to yield a single product. The addition to unsymmetrical alkenes will follow Markovnikov’s rule which states that “in the addition of HX to an alkene, the H adds to the carbon with fewer alkyl substituents and X adds to the carbon with more alkyl substituents”.
To state:
Which alkene will give 3-bromohexane as product in an addition reaction.
d)
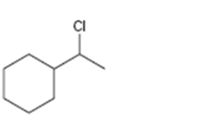
Interpretation:
The alkene that will give 1-chloroethylcyclopentane in an addition reaction is to be identified.
Concept introduction:
In addition reactions small molecules like HCl, H2O, HBr etc. add to the double bond in an alkene to yield a single product. The addition to unsymmetrical alkenes will follow Markovnikov’s rule which states that “in the addition of HX to an alkene, the H adds to the carbon with fewer alkyl substituents and X adds to the carbon with more alkyl substituents”.
To state:
Which alkene will give 1-chloroethylcyclohexane as product in an addition reaction.
Trending nowThis is a popular solution!

Chapter 7 Solutions
Organic Chemistry
- 4. What are the products from the following reaction? H3O+ کاarrow_forwardFind out what kind of reaction each alkene goes through. Figure out what the main products of each equation will be. Draw these things and name them.arrow_forwardDraw structures for the alkene (or alkenes) that gives the following reaction product.arrow_forward
- The pictured reaction shows an alkyl bromide being converted into an alkene. Choose all reagents that would produce the pictured alkene as the major product. A) NaOH/H2O B) H2O C) tBuOK/tBuOH D) EtONa/EtOHarrow_forward4) Fill in the appropriate starting materials, reagents or products in the following reactions. OH / AIC13 NO₂ Y CI / AICI,arrow_forwardDraw the alkene that would react with the reagent given to account for the product formed. ? + HCI My 3 You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. CH3 CH₂ CHOCH3 TT CI CH3 L ▼ {n [F ? ChemDoodleⓇarrow_forward
- CCH H20, H2SO4 H9SO4 CH3 Alkynes do not react directly with aqueous acid as do alkenes, but will do so in the presence of mercury(II) sulfate as a Lewis acid catalyst. The reaction occurs with Markovnikov regiochemistry, so the OH group adds to the more highly substituted carbon and the H adds to the less highly substituted carbon. The initial product of the reaction is a vinyl alcohol, also called an enol. The enol immediately rearranges to a more stable ketone via tautomerization. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions Hjö: -CH3 -CH3 H3O*arrow_forwardDraw the organic product that is expected to form when the following compound is oxidized under biological conditions. oxidation reduction 0 SH • You do not have to consider stereochemistry. You do not have to explicitly draw H atoms. If no reaction occurs, draw the organic starting material. / BO O-Sn [F ChemDoodle Previous Marrow_forwardHow do you prepare pent-2-ol from ethyne? This is alkenes and alkynes preparationarrow_forward
- Which of the structures A through D shown below will react the fastest with water? Click on a letter A to D to answer. Br Br Br H;C-Br H3C *CH B.arrow_forwardThis reaction is an example of: H H OOOOO H Halogenation Reduction Substitution Addition Oxidation H H₂SO4 concentrated heat H H SO3H H H H + H₂Oarrow_forwardWhich represents an efficient synthetic route to go from an alkane to an alkene? O elimination with NaNH2, followed by a water workup O anti-Markovnikov hydrohalogenation, followed by elimination O radical bromination, followed by elimination O hydration, followed by elimination O hydration, followed by ozonolysis of the double bondarrow_forward
