
Concept explainers
(a)
Interpretation:
The major product for the reaction of given
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Nucleophilic nature depends on the negative charge present in the molecule, the solvent in which it present and the electronegativity of the atom.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
The curved arrows are generally used to indicate the flow of electrons present in the reaction.
Carbocation: it is carbon ion that bears a positive charge on it.
Carbocation stability order:
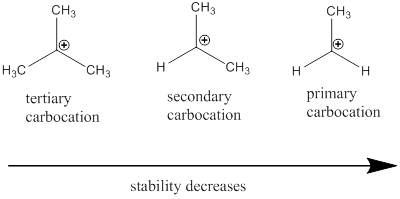
Resonance stabilization: Due to the delocalization of electrons within the molecule the overall energy becomes lower and makes that molecule more stable.
Hydroboration reaction: The reaction involves addition of
(b)
Interpretation:
The major product for the reaction of given alkene under hydro boration should be determined.
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Nucleophilic nature depends on the negative charge present in the molecule, the solvent in which it present and the electronegativity of the atom.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Chemical reaction involves bond making and breaking of two or more reactants in order to attain products from the reactants.
The curved arrows are generally used to indicate the flow of electrons present in the reaction.
Carbocation: it is carbon ion that bears a positive charge on it.
Carbocation stability order:

Resonance stabilization: Due to the delocalization of electrons within the molecule the overall energy becomes lower and makes that molecule more stable.
Hydroboration reaction: The reaction involves addition of
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Organic Chemistry (8th Edition)
- What alkylborane is formed from hydroboration of each alkene? CH3 c=CH2 b. CH2 a. C. CH3arrow_forward6. What is the major organic product obtained from the following reaction? 1. KCN 2. H30*arrow_forward7) You want to synthesize 3-methyl-2-pentene from 2-chloro-3-methylpentane. Which reagent would you use? a. HCI, heat b. NH:(aq), 25°C c. CH:CO2NA, CH:CO2H, heat d. CH3CH2ONA, CH3CH2OH, heat e. CН:CH2ОН, heatarrow_forward
- 22. Which of the following reactants will add symmetrically to alkenes? a. H20, H2SO4 O b. H2, Pt Ос. НС Od. HBrarrow_forwardIdentify the Type of Product 1-propene + H2O—> 1-propanol a. zaitsev b. markovnikov c. anti-markovnikov d. hoffmanarrow_forward2) The product of the reaction shown is which of the following? a. An alkane b. A cis alkene c. A trans alkene d. An alkyne e. The reaction fails H2 (1 atm) Pd(s), 25 °Carrow_forward
- A hydrocarbon, C6H12, on ozonolysis gives only one product which does not give silver mirror with Tollens' reagent. The hydrocarbon is ______________. a. 3-Hexene b. 2-Hexene c. 2-Methyl-2-pentene d. 2,3-Dimethyl-2-butenearrow_forwardAccording to Zaitsev’s rule, which compound will be the major product for the elimination of 2-iodobutane? A. n-butane B. butene C. butan-2-ol D. but-2-enearrow_forwardWhich of the following reagents would be used as a chemical test for the presence of alkenes and alkynes? O a. Br2 O b. H2O О с. HCl O d. CH3OHarrow_forward
- name and draw the product/s formed from the reaction of 3-methylpent-2-ene: 1. with HBr 2. with Cl2 3. H2O in the presence of H2SO4 4. ethanol in the presence of H2SO4 5. Br2, H2O 6. [1] 9-BBN; [2] H2O2, OH-arrow_forwardFor each of the following alkynes, draw the products obtained from (1) the acid-catalyzed addition of water (mercuric ion is added for part a) and from (2) hydroboration–oxidation: a. 1-butyne b. 2-butyne c. 2-pentynearrow_forwardDraw structural formulas for the alkene that gives each alcohol upon hydroborationoxidation. OH HO, b. a.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





