
(a)
Interpretation:
The ground-state electronic configuration of
Concept Introduction:
The fundamental principles that are followed to write an electronic configuration include three rules as follows:
Electron in a
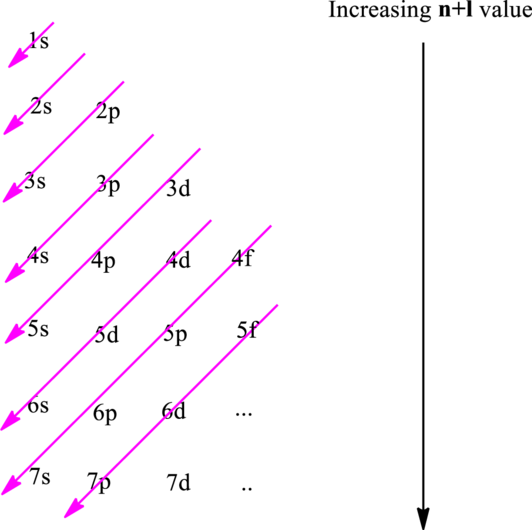
Hund’s rule suggests electrons are not allowed to be paired up until each degenerate set of orbital has got at least one electron.
Pauli Exclusion Principle states two electrons within the same orbital cannot possess same set for four possible quantum numbers.
In
(b)
Interpretation:
The ground-state electronic configuration of
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The ground-state electronic configuration of
Concept Introduction:
Refer to part (a).
(d)
Interpretation:
The ground-state electronic configuration of
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Chemical Principles: The Quest for Insight
- Draw a Lewis electron-dot symbol for (a) Ba; (b) Kr; (c) Br.arrow_forward(c) Draw the orbital diagrams and Lewis symbols to depict the formation of Na* and CI ions from the atoms. Give the formula of the compound formed. (d) The predicted bond length for HF is 109 pm (the sum of the covalent radii of H, 37 pm and F. 72 pm), however the actual bond length for HF is shorter (92 pm). It was observed that the difference between predicted and actual bond lengths becomes smalleor going down the halogen group from HF to HI Describe these observationsarrow_forwardFor many years after they were discovered, it was believed that the noble gases could not form compounds. Now we know that belief to be incorrect. A mixture of xenon and fluorine gases, confined in a quartz bulb and placed on a windowsill, is found to slowly produce a white solid. Analysis of the compound indicates that it contains 77.55% Xe and 22.45% F by mass.(a) What is the formula of the compound?(b) Write a Lewis structure for the compound.(c) Predict the shape of the molecules of the compound.(d) What hybridization is consistent with the shape you predicted?arrow_forward
- Draw a Lewis electron-dot symbol for (a) Sr; (b) P; (c) S.arrow_forwardChemical species are said to be isoelectronic if they have the same Lewis structure (regardless of charge). Consider these ions and write a Lewis structure for a neutral molecule that is isoelectronic with them. (a) CN–, (b) NH4+ (c) CO3 2–arrow_forwardDraw a Lewis electron-dot symbol for (a) As; (b) Se; (c) Ga.arrow_forward
- Testosterone is an anabolic steroid. The structure of testosterone is shown below. What is the geometry of the electron pairs at (iii) ? Hint: atom (iv) has been completed for you as an example. (iv) bond angle: 109.5°, geometry of the electron pairs: tetrahedral, shape of the bonds: tetrahedral i iii •··||||I H ····||||I ii ivarrow_forwardWrite condensed electron configurations for the following: (a) Zr; (b) V3+; (c) Mo3+.arrow_forwardTestosterone is an anabolic steroid. The structure of testosterone is shown below. What is the idealised bond angle at the indicated atom (ii) ? Hint: atom (iv) has been completed for you as an example. (iv) bond angle: 109.5°, geometry of the electron pairs: tetrahedral, shape of the bonds: tetrahedral ·····|||I H ····||||I ii ivarrow_forward
- Define the following phenomena with reference to main group compounds: (d) multicenter electron deficient bondingarrow_forwardWhich of the following bonds are polar: (a) P—O; (b) S—F; (c) Br—Br; (d) O—Cl? Which is the more electronegative atom in each polar bond?arrow_forwardIf an element is bonded to 4 other atoms and has a formal charge of +1, what group must the element be in? I know that group 3A atoms are elctron deficient, and that period 3 elements and below, except for group 3A elements like Aluminum, can expand their octet because of their available d-orbital, which may not be relevant to this problem. I don't understand this question, or why the answer would be 5A. Is it because 5A have odd valence electrons, and can form free radicals, like NO?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





