
Interpretation:
The chemical formula and the electronic configuration of phosphide ion have to be predicted.
Concept Introduction:
The fundamental principles that are followed to write an electronic configuration include three rules as follows:
Electron in a
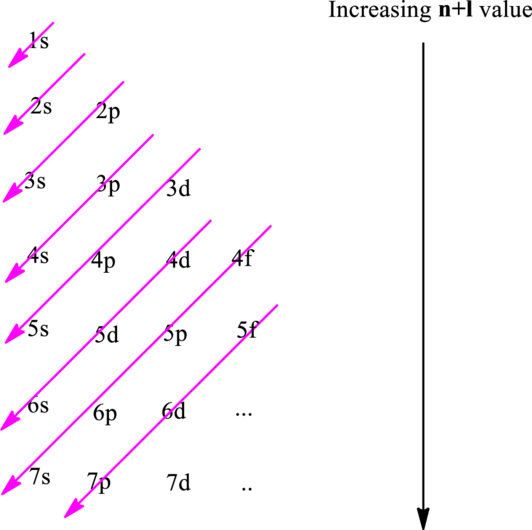
Hund’s rule suggests electrons are not allowed to be paired up until each degenerate set of orbital has got at least one electron.
Pauli Exclusion Principle states two electrons within the same orbital cannot possess same set for four possible quantum numbers.
In
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Chemical Principles: The Quest for Insight
- How does the acidity of the binary acid of an element vary as a function of the electronegativity of the element? How does this relate to the position of the element in the periodic table?arrow_forwardwhat is the formal charge on the singly bonded oxygen atoms in the lewis structure for the carbonate ion?arrow_forwardWrite the Lewis structure for carbonate ion.arrow_forward
- Draw the valence bond configuration for all the 3A, 5A, and 7A elements.arrow_forwardExplain chemically nonequivevalent hydrogen.arrow_forwardAssuming that the effect of bond energy is more important than the effect of electronegativity, the substance HI is expected to be a stronger acid than the substance HCl.arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning


