
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 15.SE, Problem 54AP
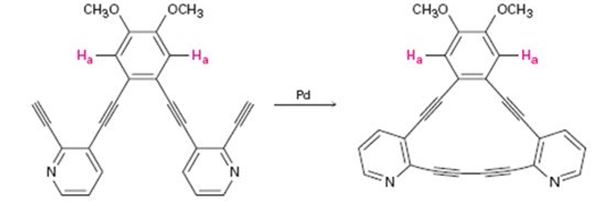
After the reaction below, the chemical shift of Ha moves downfield from 6.98 ppm to 7.30 ppm. Explain.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Explain the reaction by observing the sets of molecular collisions below.
B.
Two successive reactions, A→B and B→C, haveyields of 73% and 68%, respectively. What is the overall percentyield for conversion of A to C?
The following mechanism is proposed:
C6H5COOH ⇌ C6H5CO2H2+ (fast)
C6H5CO2H2+ ⇌ C6H5CO+ + H2O (fast)
C6H5CO+ + HX + H2O ⟶ C6H6NH2 + N2 + CO2 (slow)
The overall enthalpy change is -237.6 kJ.
Sketch a reaction-energy diagram consistent with the above mechanism and the enthalpy change. Label the enthalpy change and each transition state. Explain the appearance of your graph.
Chapter 15 Solutions
Organic Chemistry
Ch. 15.1 - Prob. 1PCh. 15.1 - Give IUPAC names for the following compounds:Ch. 15.1 - Prob. 3PCh. 15.2 - Pyridine is a flat, hexagonal molecule with bond...Ch. 15.3 - Prob. 5PCh. 15.4 - Draw the five resonance structures of the...Ch. 15.4 - Prob. 7PCh. 15.4 - Prob. 8PCh. 15.5 - Prob. 9PCh. 15.5 - Prob. 10P
Ch. 15.6 - Prob. 11PCh. 15.6 - How many electrons does each of the four nitrogen...Ch. 15.SE - Give IUPAC names for the following substances (red...Ch. 15.SE - All-cis cyclodecapentaene is a stable molecule...Ch. 15.SE - 1, 6-Methanonaphthalene has an interesting 1H NMR...Ch. 15.SE - Prob. 16VCCh. 15.SE - Azulene, an isomer of naphthalene, has a...Ch. 15.SE - Give IUPAC names for the following compounds:Ch. 15.SE - Draw structures corresponding to the following...Ch. 15.SE - Prob. 20APCh. 15.SE - Prob. 21APCh. 15.SE - Draw and name all possible aromatic compounds with...Ch. 15.SE - Propose structures for aromatic hydrocarbons that...Ch. 15.SE - Look at the three resonance structures of...Ch. 15.SE - Prob. 25APCh. 15.SE - Prob. 26APCh. 15.SE - Look at the five resonance structures for...Ch. 15.SE - Prob. 28APCh. 15.SE - 3-Chlorocyclopropene, on treatment with AgBF4,...Ch. 15.SE - Prob. 30APCh. 15.SE - Prob. 31APCh. 15.SE - Prob. 32APCh. 15.SE - Which would you expect to be most stable,...Ch. 15.SE - How might you convert 1, 3, 5, 7-cyclononatetraene...Ch. 15.SE - Calicene, like azulene (Problem 15-17), has an...Ch. 15.SE - Pentalene is a most elusive molecule that has been...Ch. 15.SE - Prob. 37APCh. 15.SE - Prob. 38APCh. 15.SE - Compound A, C8H10, yields three substitution...Ch. 15.SE - Prob. 40APCh. 15.SE - Propose structures for compounds that fit the...Ch. 15.SE - Prob. 42APCh. 15.SE - Prob. 43APCh. 15.SE - N-Phenylsydnone, so-named because it was first...Ch. 15.SE - Prob. 45APCh. 15.SE - Prob. 46APCh. 15.SE - Prob. 47APCh. 15.SE - Propose a structure for a molecule C14H12 that has...Ch. 15.SE - The proton NMR spectrum for a compound with...Ch. 15.SE - The proton NMR spectrum of a compound with formula...Ch. 15.SE - Aromatic substitution reactions occur by addition...Ch. 15.SE - Prob. 52APCh. 15.SE - Consider the aromatic anions below and their...Ch. 15.SE - After the reaction below, the chemical shift of Ha...Ch. 15.SE - Prob. 55APCh. 15.SE - Azo dyes are the major source of artificial color...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- For the arrhenius equation what are the other deviations for the formula like the ones i have to solve for t1 and k1.arrow_forward11:08 1 Question 23 of 25 Submit Construct the expression Kc for the following reaction. 2 P(s) + 5 Cl2(g) = 2 PCI;(g) 1 Drag the tiles into the numerator or denominator to form the expression. K. = 5 RESET [P] [Cl.] [PCIg] 2[P] 5[Cl_] 2[PCI;] [P]? [PCIs]? 2[P]? 5[Cl_]$ 2[PCIs]?arrow_forwardThis question has several parts that must be completed sequentially. Please use the values in the resources listed below instead of the textbook values. Derivation Calculate the equilibrium constant for the following reaction at 1224 K. 12(g) = 2 I(g) The bond length of I₂ is 2.67 Å, and the vibrational wavenumber is 214.5 cm-1. The bond dissociation energy of I₂ is 151.0 kJ.mol-¹. [Hint: To calculate the degeneracy of the iodine atom in its ground electronic state, note that there is an unpaired electron in the 5p orbital. The degeneracy is given by (2J + 1), where J, the total angular momentum, is given by the sum of the orbital angular momentum and spin angular momentum. (Assume a standard pressure of 1.00 bar and a mass of 126.90 amu for I.)] Step 1 of 11 Provide the equation needed to calculate the translational partition function for the iodine atom. (Use the following as necessary: h, kB, m, π, T, and V.) (2лmkT) (³) v V B qtrans, I = (2πmkBT) V h³ h³ Step 2 of 11 Substitute…arrow_forward
- For the following questions state whether there would be a shift to the right, left, more of a shift to the right or left, or nothing. If asked why a reaction shifts in a particular reason the answer should be kept simple because it decreases, increases, or does nothing to a specific reactant, product, or nothing to the main reaction. State what the exact reactant. For instance, the below reaction if something is added that decreases NH3, then write "the reaction shifts to the left because it decreases NH,". 1.) For the following reaction used in class [Cu(H,O),]*?(aq) + 4NH3(aq) → [Cu(NH3),]**(aq) + 4H,O(!) we know that ammonia is a weak base and its reactions with acid produces the following NH3(aq) + H*(aq) → NH,(aq) a.) If H;PO.(aq) is added what direction would the reaction shift to and why? b.) If molecular oxygen (O2) is added what direction will the reaction shift to. Note: 4NH:(aq) + 302(g) → 2N2(g) + 6H,0(!) 2.) For the following reaction that occurs in ethanol д+ 2Fe,0,(ag)…arrow_forward10:15 1 Question 7 of 25 Submit Construct the expression Kc for the following reaction. Sn(s) + 4 Cl2(g) =2 SnCl4(1) 1 Drag the tiles into the numerator or denominator to form the expression. K. = 5 RESET [SnCl.] [Cl.] [Sn] 2[SnCl4] 4[Cl2] 2[Sn] [SnCl.]? [Cl,]* [Sn]? 2[SnCl.]? 4[Cl_]* 2[Sn]?arrow_forwardFor the crystal violet reaction, why is it important that we know the initial concentration of NaOH very accurately and yet for crystal violet, we do not need to know its initial concentration?arrow_forward
- Two successive reactions, D→E and E→F, haveyields of 48% and 73%, respectively. What is the overall percentyield for conversion of D to F?arrow_forward下午9:55 6月20日 週一 < S T Q + a) I, II, III, IV b) III, IV, II, I c) II, I, III, IV d) IV, III, II, I 3. Wh 3. Which compound undergoes the fastest exchange of hydrogen for deuterium when treated with D₂0 and a trace of OD? (Sec. 14.2) H₂C CH3 0 *xe a) 191 50% U 58 91 v ✪arrow_forwardFor the reaction N2(9) + O2(9) = 2NO(g) classify each of the following actions by whether it causes a leftward shift, a rightward shift, or no shift in the direction of the reaction. Drag the appropriate items to their respective bins. • View Available Hint(s) Reset Help halve Po2 halve double PN2 double halve PN2 double Po2 PNO PNO Leftward shift Rightward shift No shiftarrow_forward
- Chemistry of Fire Extinguishing Agents: Review and answer the below scenerios. a. The OSHA regulation at 29 C.F.R. §1910.162(b)(5) requires employers to provide a predischarge-employee alarm system to alert employees if a carbon dioxide concen-tration of 4% or greater is released from a fixed extinguishing system in a workplace. What is the most likely reason OSHA enacted this regulation? b. At 14 C.F.R. §121.265, FAA allows carbon dioxide to be used as a fire extinguish-ing agent onboard aircraft, provided that provisions are made to prevent harmful concentrations from entering any personnel compartment. Use Table 5.6 to deter-mine whether adverse health effects are likely to be experienced by a crew member who is confined to a compartment in which the air contains 8, carbon dioxide. c. The chemical formula of methyl bromide is CH3Br. Why does methyl bromide per-form effectively as a fire extinguishing agent? d. A potential risk of inhalation toxicity may occur to individuals that…arrow_forwarda) Explain the differences between general acid/base catalysed reaction and specific acid/base catalysed reaction b) Sketch a plot of log k against pH for specific acid catalysed reaction and general acid catalysed reaction and explain.arrow_forwardCalculate the concentration of N2O5 in the vessel 9.90 seconds later. Round to two dig figs.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY