
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14, Problem 14.30SP
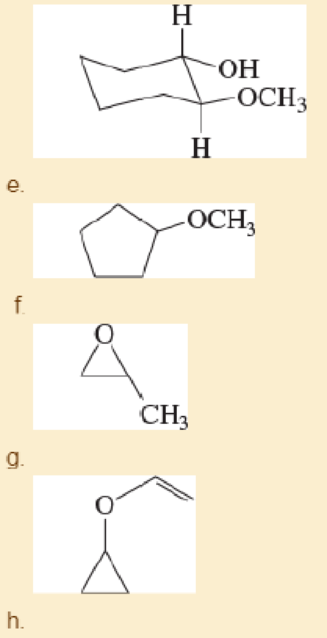
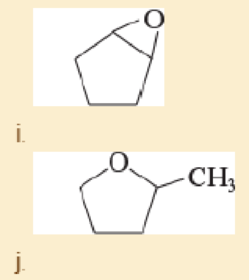
Give common names for the following compounds.
- a. (CH3)2CHOCH(CH3)CH2CH3
- b. (CH3)3COCH2CH(CH3)2
- c. PhOCH2CH3
- d. ClCH2OCH2CH2CH3
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw the products formed when CH3CH2C ≡ C−Na+reacts with each compound.
a. CH3CH2CH2Brb. (CH3)2CHCH2CH2Clc. (CH3CH2)3CCld. BrCH2CH2CH2CH2OHe. ethylene oxide followed by H2Of. propene oxide followed by H2O
Reagents
a. C6H5CHO
b. NaOH, ethanol
h. BrCH2CH=CH2
i.
Na* OEt, ethanol
j. Br2, H*
k. K* t-BuO
c. Pyrrolidine, cat. H*
d. H2C=CHCN
e. H3O*
f.
I. CH2(CO2ET)2
-CH2CH2CN
LDA
m. heat
g. ELOC(=0)CO2ET
Select reagents from the table to synthesize this compound from cyclopentanone.
Enter the letters of the chosen reagents, in the order that you wish to use them, without spaces or punctuation (i.e. geda).
33. What is each compound's systematic name?
a. CH₂C=CCH₂CHCH, b. CH₂C=CCH₂CHCH₂
Br
CH₂CH₂CH₂
CH₁
c. CH₂C=CCH₂CCH, d. CH₂CHCH₂C=CCHCH
CH,
CH,
Chapter 14 Solutions
Organic Chemistry (9th Edition)
Ch. 14.2C - Rank the given solvents in decreasing order of...Ch. 14.2D - Prob. 14.2PCh. 14.2D - In the presence of 18-crown-6, potassium...Ch. 14.3B - Give a common name (when possible) and a...Ch. 14.3C - Prob. 14.5PCh. 14.3C - Name the following heterocyclic ethers.Ch. 14.4 - Propose a fragmentation to account for each...Ch. 14.5 - Prob. 14.8PCh. 14.5 - Prob. 14.9PCh. 14.6 - Prob. 14.10P
Ch. 14.7 - Explain why bimolecular condensation is a poor...Ch. 14.7 - Prob. 14.12PCh. 14.7 - Prob. 14.13PCh. 14.8 - Prob. 14.14PCh. 14.8 - Prob. 14.15PCh. 14.8 - Prob. 14.16PCh. 14.10A - Prob. 14.17PCh. 14.10A - Prob. 14.18PCh. 14.10B - Prob. 14.19PCh. 14.11B - Show how you would accomplish the following...Ch. 14.11B - Prob. 14.21PCh. 14.12 - Prob. 14.22PCh. 14.12 - Prob. 14.23PCh. 14.12 - Prob. 14.24PCh. 14.13 - Prob. 14.25PCh. 14.13 - Prob. 14.26PCh. 14.14 - Prob. 14.27PCh. 14.15 - Give the expected products of the following...Ch. 14 - Write structural formulas for the following...Ch. 14 - Give common names for the following compounds. a....Ch. 14 - Give IUPAC names for the following compounds. a....Ch. 14 - Glycerol (propane-1,2,3-triol) is a viscous syrup...Ch. 14 - Prob. 14.33SPCh. 14 - Show how you would make the following ethers,...Ch. 14 - (A true story.) An inexperienced graduate student...Ch. 14 - Prob. 14.36SPCh. 14 - a. Show how you would synthesize the pure (R)...Ch. 14 - a. Predict the values of m/z and the structures of...Ch. 14 - The following reaction resembles the...Ch. 14 - Prob. 14.40SPCh. 14 - Prob. 14.41SPCh. 14 - Prob. 14.42SPCh. 14 - Give the structures of the intermediates...Ch. 14 - Prob. 14.44SPCh. 14 - Show how you would synthesize the following ethers...Ch. 14 - Prob. 14.46SPCh. 14 - Prob. 14.47SPCh. 14 - Prob. 14.48SPCh. 14 - An acid-catalyzed reaction was carried out using...Ch. 14 - Propylene oxide is a chiral molecule. Hydrolysis...Ch. 14 - Prob. 14.51SPCh. 14 - Prob. 14.52SPCh. 14 - Prob. 14.53SPCh. 14 - Prob. 14.54SPCh. 14 - In 2012, a group led by Professor Masayuki Satake...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which structure is a meso compound? A. В. С. D. CH3 CH 3 CH3 CH 3 H--C1 H--C1 H--C1 H--C1 cl-- - CH3 Cl--H H--C1 Cl--H CH 3 CH2CH3 CH2CH3arrow_forwardProvide the IUPAC names for the following compounds. Compound IUPAC Name 1. H3Ç CH3 H2C=CHCHCCH3 CHa 2. CH3 CH3CH2CH=CH2CH3 3. CH3 CH3CH=CHCHCH=CHCHCH3 CH3 4. CH3CHCH2CH3 CH3CH2CH2CH=CHCHCH2CH3 5. „CH3 CH3arrow_forwardChapter 1 1. Functional Group Interconversion. Show how each of the following compounds can be prepared from the given starting material provided. b. C. d. *e. *f. CO₂H по дон (R) HO OH THPO OAC CH2NH2 CH3 H H Ph → provala (S), OH THE THINGS TO THE TEarrow_forward
- a. The following compounds have the same molecular formula as benzene. How many monobrominated products could each form? 1. HC‚CC‚CCH2CH3 2. CH2“CHC‚CCH“CH2 b. How many dibrominated products could each of the preceding compounds form? (Do not include stereoisomers.) c. How many dibrominated products could each of the compounds form if stereoisomers are included?arrow_forwardCis-but-2-ene has a different set of properties compared to trans-but-2-ene. the melting points of these two compounds is influence primarily by what factor? a. Dipole moment b. Hydrogen Bonding c. Symmetry of molecule d. Van de Waals interaction Which electrophilic aromatic substitution reaction is described when aniline is transformed into para-aminotolouene? a. Halogenation b. Nitration c. Oxidation d. Friedel Crafts alkylation The following hydrocarbons will exhibit electrophilic reactions (addition or substitution) involving halogens except: a. ethyne b. ethene c. ethane d. benzenearrow_forwardName the following: a. BrCH2CH2C CCH3 b. CH3CH2CHBrC=CCH2CHClCH3 c. CH3OCH2C=CCH2CH3 d. CH3CH2CHCH2CH2CH3C=CHarrow_forward
- A. Naming Structures 1. Use the JUPAC rules to name the following structures. CH CH = CHCHCH, b. CH3 a. CH3 CH2 CH3 CH-CH сн, CH3CHCH2CH =CHCCH, CH3 2. Use IUPAC rules to name the following structures. CH,CH3 b. CH3 CH3 CH3 CH,CH, CH3CH, В. B. Draw the structures of the following cycloalkanes 1. 2,3-dimethyl-5-propyldecane 2. 3,4,5-triethyloctane 3. 1-ethyl-3-propylcyclopentane 4. 1,2,2,4-tetramethylcyclohexanearrow_forwardName the following compounds: a. CH3OCH2CH3 b. CH3OCH2CH2CH3 c. CH3CH2CH-CH3NH2 d. CH3CH2CH2CH2OH e. CH3CH-CH3CH2Br f. CH3CH2CHCH3Clarrow_forward9. Which compounds react together to form an ester? CH3CH₂OH CH3CH₂COOH II. CH3CH CHO IV. CH3CH₂COCH3 I. III. I and IV only II and III only I and III only D) Both I and II and I and IV would produce esters. A) B) C)arrow_forward
- Give the IUPAC name for each compound. OH a. CH3CH(CH₂)4CH3 (select) OH (CH3CH₂)2CHCHCH₂CH3 (select) b. C. d. CH3 OH (select) (select) (select) OH (select) (select) (select)arrow_forward6. What is the product? CH3 осно A. C. A and B A. A B. B C. C CH3 B. DBN CH₂arrow_forward10. Give the IUPAC name for each of the following compounds. a) CH3CH2CH₂CH₂CHCICH2CH₂CH3 CI F b) CH3CH₂ CH3CHCH₂CHCH 3 I F CH3CH₂CHCH₂CHCH₂CH3 HCH₂CH3 d)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY