
Organic Chemistry
5th Edition
ISBN: 9780078021558
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13, Problem 13.23P
What major IR absorptions are present above
a. 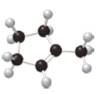 b.
b. 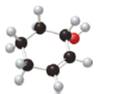
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
B.19 What major IR absorptions are present above 1500 cm ¹ for each
compound?
a.
b.
C.
d.
OH
OH
What major IR absorptions are present above 1500 cm- for each compound?
а.
b.
What major IR absorptions are present above 1500 cm−1 for each compound?
Chapter 13 Solutions
Organic Chemistry
Ch. 13 - What is the mass of the molecular ion formed from...Ch. 13 - Prob. 13.2PCh. 13 - Use the following information to propose a...Ch. 13 - Prob. 13.4PCh. 13 - What molecular ions would you expect for the...Ch. 13 - The mass spectrum of 2,3-dimethylpentane also...Ch. 13 - The base peak in the mass spectrum of 2, 2,...Ch. 13 - (a) What mass spectral fragments are formed by ...Ch. 13 - What cations are formed in the mass spectrometer...Ch. 13 - The low-resolution mass spectrum of an unknown...
Ch. 13 - Benzene, toluene, and p-xylene BTX are often added...Ch. 13 - Prob. 13.12PCh. 13 - Prob. 13.13PCh. 13 - Prob. 13.14PCh. 13 - Prob. 13.15PCh. 13 - How do the IR spectra of the isomers cyclopentane...Ch. 13 - Problem 13.17 How do the three isomers of...Ch. 13 - Problem 13.18 What functional groups are...Ch. 13 - Problem-13.19 What are the major IR absorptions in...Ch. 13 - Problem-13.20 What are the major IR absorptions in...Ch. 13 - Problem-13.21 Which of the following possible...Ch. 13 - Problem-13.22 Propose structures consistent with...Ch. 13 - 13.23 What major IR absorptions are present above ...Ch. 13 - Problem-13.24 The mass spectrum of the following...Ch. 13 - Prob. 13.25PCh. 13 - Which compound gives a molecular ion at m/z= 122,...Ch. 13 - Propose two molecular formulas for each molecular...Ch. 13 - Propose four possible structures for a hydrocarbon...Ch. 13 - Problem-13.29 What is the molecular formula for...Ch. 13 - Problem-13.30 Propose a molecular formula for rose...Ch. 13 - 13.31 Match each structure to its mass spectrum
Ch. 13 - 13.32 Propose two possible structures for a...Ch. 13 - 13.33 What cations are formed in the mass...Ch. 13 - 13.34 and have the same molecular ion in the...Ch. 13 - 13.35 For each compound, assign likely...Ch. 13 - Prob. 13.36PCh. 13 - 13.37 Propose a structure consistent with each...Ch. 13 - 13.38 A low-resolution mass spectrum of the...Ch. 13 - 13.39 Primary alcohols often show a peak in their...Ch. 13 - 13.40 Like alcohols, ethers undergo α cleavage by...Ch. 13 - 13.41 Which of the highlighted bonds absorbs at...Ch. 13 - 13.42 What major IR absorptions are present above ...Ch. 13 - 13.43 How would each of the following pairs of...Ch. 13 - 13.44 Morphine, heroin, and oxycodone are three...Ch. 13 - 13.45 Reduction of cyclohex-2-enone can yield...Ch. 13 - Prob. 13.46PCh. 13 - 13.47 Match each compound to its IR spectrum
Ch. 13 - 13.48 Propose possible structures consistent with...Ch. 13 - A chiral hydrocarbon X exhibits a molecular ion at...Ch. 13 - 13.50 A chiral compound has a strong absorption...Ch. 13 - 13.51 Treatment of benzoic acid with followed by...Ch. 13 - 13.52 Treatment of benzaldehyde with in aqueous ...Ch. 13 - Prob. 13.53PCh. 13 - 13.54 Reaction of 2-methylpropanoic acid with ...Ch. 13 - 13.55 Reaction of pentanoyl chloride with lithium...Ch. 13 - Prob. 13.56PCh. 13 - 13.57 Treatment of anisole with and forms P,...Ch. 13 - 13.58 Reaction of with forms compound ,...Ch. 13 - Problem-13.59 The carbonyl absorption of an amide...Ch. 13 - Prob. 13.60PCh. 13 - Problem-13.61 Explain why a ketone carbonyl...Ch. 13 - 13.62 Oxidation of citronellol, a constituent of...
Additional Science Textbook Solutions
Find more solutions based on key concepts
1. What did each of the following scientists contribute to our knowledge of the atom?
a. William Crookes
b. E...
Chemistry For Changing Times (14th Edition)
Real walls are never totally adiabatic. Use your experience to order the following walls in increasing order wi...
Thermodynamics, Statistical Thermodynamics, & Kinetics
The active ingredient in Tylenol and a host of other over-the-counter pain relievers is acetaminophen (C8H9NO2)...
Chemistry: Atoms First
141. Design a device that uses as electrochemical cell to determine amount of
in a sample water Describe, in...
Chemistry: Structure and Properties (2nd Edition)
2. Why shouldn’t you work in a laboratory by yourself?
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- IR spectroscopy does not distinguish very well betweenisomers I and J. Explain why. How could UV–vis spectroscopy be used to distinguish between themarrow_forwardwhat compound has this given IR Spectra? 80 2725 2827 40 2976 1731 of 4000 2000 Wavenumbers (cm-1) O A. butanol B. butanoic acid O C. butanone O D. butyraldehyde %Transmittancearrow_forwardWhat major IR absorptions are present above 1500 cm−1 for attached compound ?arrow_forward
- How does the operating frequency in NMR spectroscopy compare with the operating frequency in IR and UV/Vis spectroscopy?arrow_forwardThis is the 13C spectrum for an X compound with molecular formula C7H12O4. The substance is not soluble in NaHCO3 and has a stretch at 1740cm-1 on the IR spectrum. What is the structure of X?arrow_forwardPropose possible structures consistent with each set of data. Assume each compound has an sp3 hybridized C—H absorption in its IR spectrum, and that other major IR absorptions above 1500 cm−1 are listed. a.a compound having a molecular ion at 72 and an absorption in its IR spectrum at 1725 cm−1 b. a compound having a molecular ion at 55 and an absorption in its IR spectrum at −2250 cm−1 c.a compound having a molecular ion at 74 and an absorption in its IR spectrum at 3600−3200 cm−1arrow_forward
- What are the major IR absorptions in the functional group region for each compound?arrow_forwardPropose possible structures consistent with each set of data. Assume each compound has an sp3 hybridized C – H absorption in its IR spectrum, and that other major IR absorptions above 1500 cm are listed. a. a compound having a molecular ion at 72 and an absorption in its IR spectrum at 1725 cm−1 b. a compound having a molecular ion at 55 and an absorption in its IR spectrum at ~2250 cm−1 c. a compound having a molecular ion of 74 and an absorption in its IR spectrum at 3600–3200 cm−1arrow_forwardWhat are the notable peaks in the IR spectra of each molecule?arrow_forward
- Choose the compound with the most intense IR signal at -1700 cm1. CI .CI .CI CI A B Darrow_forwardIdentify the important absorption peaks in the following IR spectra and describe the appearance of the important absorption peaks in the IR Spectra. No cursive writing please. Thank youarrow_forwardPropose possible structures consistent with each set of data. Assume each compound has an sp hybridized C-H absorption in its IR spectrum, and that other major IR absorptions above 1500 cm1 are listed. a. a compound having a molecular ion at 72 and an absorption in its IR spectrum at 1725 cm-1 b. a compound having a molecular ion at 55 and an absorption in its IR spectrum at -2250 cm- c. a compound having a molecular ion of 74 and an absorption in its IR spectrum at 3600-3200 cm"arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY