
Interpretation:
The product of photochemical cyclization reaction of
Concept Introduction:
>The compounds that have the same molecular formula of atoms but the structure of the compounds are different for the positions of atoms are called isomers.
>In double bond or cyclic compounds, if two same
If the two functional groups are present on the different sides of the double bond or cyclic compound, the given compound can be labeled as Trans.
>Cis-trans isomerism exists in the compounds in which similar groups are present on the adjacent carbon atoms.
>The thermal electrocyclic reaction having
The photochemical electrocyclic reaction having
The electrocyclic reactions are initiated by heat or light and are stereospecific in nature.
Answer to Problem 1PP
Solution:
The product of the reaction is given below:
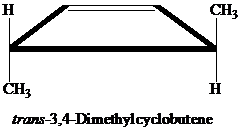
Explanation of Solution
The compound
Thus, the photochemical cyclization reaction of

The product of the reaction is
Want to see more full solutions like this?
Chapter D Solutions
Organic Chemistry, 12e Study Guide/Student Solutions Manual
- Problem 3 of 16 Draw the products of this reduction of a ketone with sodium borohydride. 1) NaBH4 49 Use a dash or wedge bond to indicate the stereochemistry of substituents on asymmetric centers, where applicableIgnore any inorganic byproducts. 2) HCI/H₂O Submit Select to Drawarrow_forwardWhat reagents are required to carry out the following reaction (select all that apply)? a HNO3 b H2SO4 c Cl2 d Br2 e H2O f CH3COCl g AlCl3 h FeCl3 i FeBr3 j CH3CHClCH3arrow_forwardProblem Addition of HCl to 1-isopropylcyclohexene yields a mixture of two products, one of which is the rearranged product. Propose a mechanism for the rearranged product shown below, showing the structures of the intermediates and using curved arrows indicate electron flow in each step. + HCI CI 56arrow_forward
- PROBLEM Predict the product(s) and provide the mechanism for each of the 18-28 following reactions. (I PROBLEM Predict the product(s) and provide the mechanism for each of the 18-28 following reactions. (a) (b) H. CH3 HBr ? ether CH3 H30+ ?arrow_forwardProblem 14 of 20 KOtBu 34 Draw the product of the E2 reaction shown below. Include the correct stereochemistry. Ignore any inorganic byproducts. Draw the E2 Product Submitarrow_forwardProblem 11 of 32 Draw the product of the substitution reaction shown below. Ignore the inorganic byproducts. CH3CH₂I DMSO OO (+) Submit Naarrow_forward
- For each reaction below, provide the product(s) that would form and indicate whether the substitution reaction would occur by an SN1 , SN2, E1, or E2 mechanism. Draw all constitutional and/or stereoisomers that would form in each reaction. I need help with this practice problem, thank you!arrow_forwardPractice Problems on SN1, SN2, El & E2 - 1. Describe the following chemical reactions as SN1, SN2, E1 & E2. Draw a curved arrow mechanism for each reaction. KCN CN DMSO C.N NaOH Br H2O, heat .. Br H20 Y.... OH CH;CH2O°Na* ethanol NaSH DMSO SH КОН Но Br DMSO SN NaNH2 OTs NH3 NH3 くTSO H2N TsO. CH3 Nal CH3 ... CH3 acetone CH3arrow_forwardProblem 5 of 44 Curved arrows are used to illustrate the flow of electrons. Using the provided starting structure, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. Then draw the organic product of this reaction. Include all lone pairs in the structures. Ignore inorganic byproducts and counterions. NECO Submit CI Select to Add Arrows NaCN Na Ⓒ THFarrow_forward
- Predict the major product of the following reactions. The formulae of the products are given in bold. Please answer d,e,f,garrow_forwardProblem 3 Propose a reasonable biosynthesis of Fenchone from geranylpyrophosphate (+-Fenchonearrow_forward1. For the following electrophilic addition reactions, predict the major products. No mechanisms necessary. PROBLEM SET H-Br H-CI H-CIarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





