
Concept explainers
Practice Problem C.1
(a) Write structural formulas for portions of the chain of the atactic, syndiotactic, and iso-tactic forms of polystyrene (see Practice Problem 10.15). (b) If solutions were made of each of these forms of polystyrene, which solutions would you expect to show optical activity?
Interpretation:
The structural formula for the atactic, syndiotactic, and isotactic forms of polystyrene is to be determined.
Concept introduction:
>The polymer, on the basis of stereochemistry around the chiral center, is classified as atactic, syndiotactic, and isotactic.
>A polymer in which the stereochemistry at the chiral centre is random is said to be the atactic polymer.
>A polymer in which the stereochemistry at the chiral centre alternates regularly, from one side to the other, on the chain is said to be the syndiotactic polymer.
>A polymer in which the stereochemistry of all chiral centres is the same is said to be the isotactic polymer.
>The polystyrene is the aromatic hydrocarbon polymer of the monomer styrene.
>A molecule is considered optically-active if it contains an achiral center and its mirror image is non-superimposable.
>The molecules which are non-superimposable or not identical with their mirror images are known as chiral molecules.
>A pair of two mirror images which are non-identical is known as enantiomers which are optically active.
>The objects or molecules which are superimposable with their mirror images are achiral objects or molecules and these objects have a centre of symmetry or plane of symmetry.
>The achiral compounds in which plane of symmetry is present internally and consists of chiral centres are known as meso compounds, but they are optically inactive.
>Diastereomers are the stereoisomers that are not mirror images of each other and are not superimposable on each other.
>They possess different physical as well as chemical properties, because of difference in orientations.
Answer to Problem 1PP
Solution:
(a) The structural formula for atactic, syndiotactic, and isotactic forms of polystyrene is as:
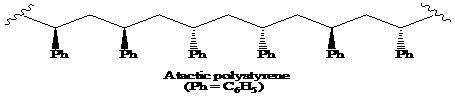
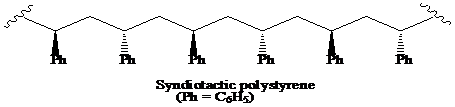
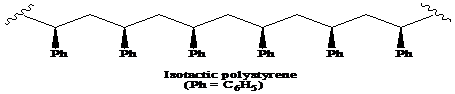
(b) The solution of isotactic polystyrene shows optical activity.
Explanation of Solution
a) The structural formula for portion of the chain of the atactic, and iso-tactic forms of polystyrene.
On the basis of arrangement of substituents on the chiral centre in the chain, the polymer is classified into atactic, syndiotactic, and isotactic.
A polymer in which the stereochemistry at the chiral centre is random is said to be the atactic polymer.
A polymer in which the stereochemistry at the chiral centre alternates regularly, from one side to the other, on the chain is said to be the syndiotactic polymer.
A polymer in which the stereochemistry of all chiral centres is the same is said to be the isotactic polymer.
Thus, the structural formula for the atactic form of polystyrene is as:
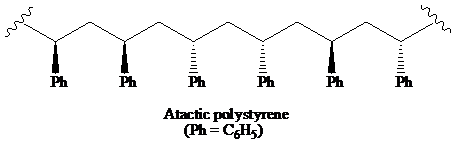
Thus, the structural formula for the syndiotactic form of polystyrene is as:

Thus, the structural formula for the isotactic form of polystyrene is as:
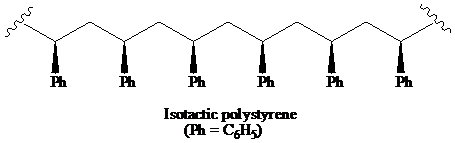
b) The solution that expected to show optical activity.
A molecule is considered optically-active if it contains an achiral center and its mirror image is non-superimposable. Among the solutions of atactic, syndiotactic, and isotactic forms of polystyrene, the solution of isotactic polystyrene rotates the plane-polarized light and its mirror image is non-superimposable. Thus, the isotactic polystyrene shows optical activity.
Therefore, the solution of isotactic polystyrene shows optical activity.
Want to see more full solutions like this?
Chapter C Solutions
Organic Chemistry
Additional Science Textbook Solutions
Living By Chemistry: First Edition Textbook
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
General, Organic, and Biological Chemistry (3rd Edition)
Chemistry
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
- Which of these structures fit the following descriptions? Select all that are correct.arrow_forwardGive the structural formulas of the compound in the following problem. Explain how you arrived at each answerarrow_forwardWould you expect Octane (C8H18 -- line-angle formula shown below) to be soluble in water? Briefly explain.arrow_forward
- Instruction: Imagine that you are performing experiments pertaining to organic chemistry. Second, follow the procedures given. Lastly, answer the questions given below. OBJECTIVES- Demonstrate the process of salting out. MATERIALS Distilled water (Absolute, green cover)Sodium Chloride (NaCl, Salt)Isopropyl Alcohol (C3H8O)Medium-sized beadsFunnelPlastic Bottlearrow_forwardHow to approach problem, Which one of the following compounds has not so far been found to exist? a) K2[CuO4] b) K2[CrO4] c) Fe3O4] d) OsO3F2arrow_forwardUsing IUPAC nomemclature rules name and provide molecular formula. Is this a constitutional or structural isomer? Name: Molecular Formula:arrow_forward
- Branched compounds are said to have weaker intermolecular forces of attraction. Therefore, between pentane, a straight-chained compound and 2-methylbutanol which exhibits branching at carbon 2, the former has stronger IMFA. (hint: look at the structure of the two molecules) True or Falsearrow_forwardName using IUPACarrow_forwardChiral compounds have a so-called asymmetric carbon atom in the molecular structure, and are opticalactive. Study the following organic molecule containing two −? − ? – groups (thiol) andwhich is a medicine against various types of metal poisoning, including against arsenic, mercury and lead: Image Based on this skeletal structure, judge whether this molecule is chiral. Give reasons for your answer.Suggest an IUPAC name for the molecule that takes into account all the given functional groups.arrow_forward
- Each of the following names is wrong. Draw structuresbased on them, and correct the names:(a) 3,3-dimethylbutane(b) 1,1,1-trimethylheptane(c) 1,4-diethylcyclopentane(d) 1-propylcyclohexanearrow_forwardI don’t understand, what is the difference between conformational structures of a molecule and different structural isomers? Could you please show an example as well? Thanksarrow_forwardName the following compounds with the type of name indicated. (Use isobutyl etc. with IUPAC, as needed)arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





