
Interpretation:
The required volume of stock solution to prepare the given volumes and concentrated final solutions are should be calculated by using given representation diagram.
Concept introduction:
Dilution:
- Dilution is the process, which is used to prepare a low concentrated solution from high concentrated solution.
Volumetric principle:
- In the dilution process, the relationship between initial and final concentrations and volumes of solutions are given in the volumetric equation and it is,
Explanation of Solution
To calculate the required volumes of stock solutions to prepare the final diluted solutions
The given diagram is,
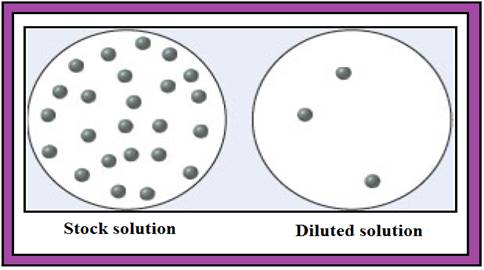
Diagram.1
To calculate the required volume of stock solution to prepare
From the given diagram (Diagram.1) the stock solution contains 24 numbers of solute particles and diluted solution contains 3 numbers of solute particles.
Consider a unit of stock solution contains 24 numbers of solute particles and a unit of diluted solution contains 3 numbers of solute particles.
The numbers of solute particles and final volume are plugged in the above equation to give required volume of stock solution to prepare
The required volume of stock solution to prepare
The required volume of stock solution to prepare
From the given diagram (Diagram.1) the stock solution contains 24 numbers of solute particles and diluted solution contains 3 numbers of solute particles.
Consider a unit of stock solution contains 24 numbers of solute particles and a unit of diluted solution contains 3 numbers of solute particles.
The numbers of solute particles and final volume are plugged in the above equation to give required volume of stock solution to prepare
The required volume of stock solution to prepare
The required volume of stock solution to prepare
From the given diagram (Diagram.1) the stock solution contains 24 numbers of solute particles and diluted solution contains 3 numbers of solute particles.
Consider a unit of stock solution contains 24 numbers of solute particles and a unit of diluted solution contains 3 numbers of solute particles.
The numbers of solute particles and final volume are plugged in the above equation to give required volume of stock solution to prepare
The required volume of stock solution to prepare
The required volume of stock solution to prepare the given volumes and concentrated final solutions are calculated by using given representation diagram.
Want to see more full solutions like this?
Chapter 9 Solutions
Chemistry: Atoms First
- ssume a highly magnified view of a solution of HCI that allows you to “see” the HCl. Draw this magnified view. If you dropped in a piece of magnesium, the magnesium would disappear, and hydrogen gas would he released. Represent this change using symbols for the elements, and write the balanced equation.arrow_forwardWhat kinds of solute particles are present in a solution of an ionic compound? Of a molecular compound?arrow_forwardCalcium carbonate, CaCO3, can be obtained in a very pure state. Standard solutions of calcium ion are usually prepared by dissolving calcium carbonate in acid. What mass of CaCO3 should be taken to prepare 500. mL of 0.0200 M calcium ion solution?arrow_forward
 Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning





