
Concept explainers
Write Lewis dot symbols for the following atoms and ions: (a) I, (b) I−, (c) S, (d) S2−, (e) P, (f) P3−, (g) Na, (h) Na+, (i) Mg, (j) Mg2+, (k) Al, (l) Al3+, (m) Pb, (n) Pb2+.
(a)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
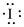
(b)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
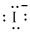
(c)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
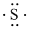
(d)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
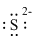
(e)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
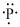
(f)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
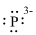
(g)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
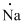
(h)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
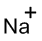
(i)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
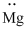
(j)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
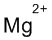
(k)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
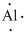
(l)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
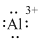
(m)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
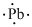
(n)
Interpretation:
Lewis dot symbol for
Concept Introduction:
Lewis dot symbol represents the number of valence electrons of an atom or constituent atoms bonded in a molecule. Each dot corresponds to one electron.
Explanation of Solution
Electronic configuration of
Want to see more full solutions like this?
Chapter 9 Solutions
ALEKS 360; 18WKS F/ GEN. CHEMISTRY >I<
- Provide an explanation for the way the following formula Ca5(PO4)3OH is written and state the name of all the ions involved. (Explain what the subscripted numbers indicate, why is PO4 in brackets whereas OH isn’t?)arrow_forward3. Use Lewis symbols to diagram the reactions between (a) Ca and Br, (b) Al and O, and (c) K and S.arrow_forwardWrite the empirical formula of at least four binary ionic compounds that could be formed from the following ions: 2+ Ca, Pb**, Br, o²- 0,0,..arrow_forward
- Write the empirical formula for at least four ionic compounds that could be formed from the following ions: + Fe³+, SO², PO¾¯, NH 0,0,... X Śarrow_forward3. How many valence electrons are in a C atom? an Cl atom? (a) Write the full Lewis structure for the CH2C2 molecule. Show all lone pairs. Pert (b) Write the geometric structure for the CH2CI2 molecule and name the geometry. (c) Determine if the CH2CI2 molecule has polar or nonpolar bonds using the following electronegativity: C = 2.5, H = 2.1, CI = 3.0. Re-draw the geometric structure below, but this time include partial charges and dipole arrows for any polar bonds present. Justify your assignments for polar or nonpolar bonds.arrow_forwardWrite the empirical formula for at least four ionic compounds that could be formed from the following ions: Pb, C,H,0,, Fe, Cio,arrow_forward
- Write the empirical formula for at least four ionic compounds that could be formed from the following ions: 3+ 4+ Fe³+, Pb²+, CN¯¯, Cro 0 0,0,... × Śarrow_forwardConsider the following compounds: BeCl 2 , MgBr 2 , and SrBr 2 . Answer the following questions based on expected periodic trends: (a) Which is expected to have the shortest ionic bonds? (b) Which is expected to have the highest lattice energy? (c) Which is expected to have the lowest melting point?arrow_forwardWrite the empirical formula of at least four binary ionic compounds that could be formed from the following ions: Fe, AI", F, 0- 0.0.arrow_forward
- Write the empirical formula for at least four ionic compounds that could be formed from the following ions: 4+ CH,CO,, CN, Pb ", NHarrow_forwardWrite the empirical formula for at least four ionic compounds that could be formed from the following ions: 4+ MnO, NH, Pb*, BrO, O0,..arrow_forwardWrite the empirical formula for at least four ionic compounds that could be formed from the following ions: Fe3+, C₂H₂O2, NH, Cro 0,0,...arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





