
What
results.
a. 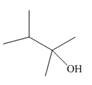 b.
b. 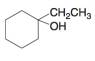 c.
c. 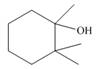 d.
d.
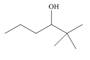
(a)
Interpretation: The alkenes formed by the treatment of given alcohol with
Concept introduction: Alcohols undergo dehydration reaction in the presence of strong acids like
Answer to Problem 9.48P
The alkenes formed by the treatment of given alcohol with
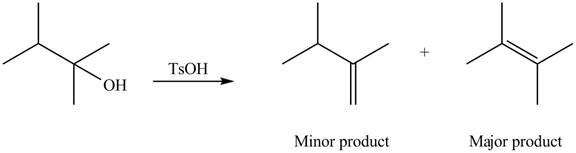
Explanation of Solution
The given alcohol contains two
Alcohols undergo dehydration reaction in the presence of strong acids like
Thus, the alkenes formed by the treatment of given alcohol with
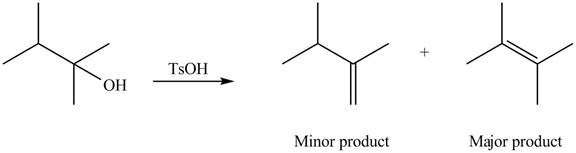
Figure 1
The alkenes formed by the treatment of given alcohol with
(b)
Interpretation: The alkenes formed by the treatment of given alcohol with
Concept introduction: Alcohols undergo dehydration reaction in the presence of strong acids like
Answer to Problem 9.48P
The alkenes formed by the treatment of given alcohol with
Explanation of Solution
The given alcohol contains two
Alcohols undergo dehydration reaction in the presence of strong acids like
Thus, the alkenes formed by the treatment of given alcohol with

Figure 2
The alkenes formed by the treatment of given alcohol with
(c)
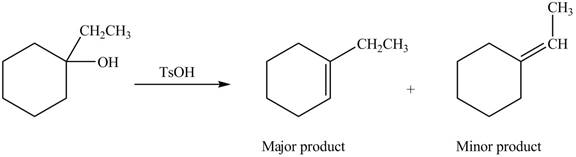
Interpretation: The alkenes formed by the treatment of given alcohol with
Concept introduction: Alcohols undergo dehydration reaction in the presence of strong acids like
Answer to Problem 9.48P
The alkenes formed by the treatment of given alcohol with
Explanation of Solution
The given alcohol contains three
Alcohols undergo dehydration reaction in the presence of strong acids like
Thus, the alkenes formed by the treatment of given alcohol with
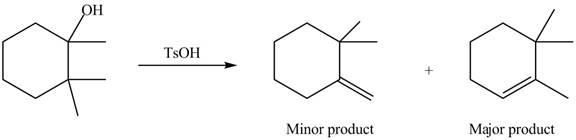
Figure 3
The alkenes formed by the treatment of given alcohol with
(d)
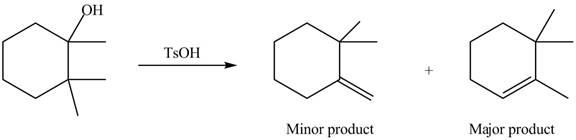
Interpretation: The alkenes formed by the treatment of given alcohol with
Concept introduction: Alcohols undergo dehydration reaction in the presence of strong acids like
Answer to Problem 9.48P
The alkene formed by the treatment of given alcohol with
Explanation of Solution
The given alcohol contains two
Alcohols undergo dehydration reaction in the presence of strong acids like
The alkenes formed by the treatment of given alcohol with
(e)
Interpretation: The alkenes formed by the treatment of given alcohol with
Concept introduction: Alcohols undergo dehydration reaction in the presence of strong acids like
Answer to Problem 9.48P
The alkenes formed by the treatment of given alcohol with
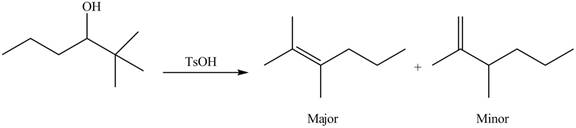
Explanation of Solution
The given alcohol contains two
Alcohols undergo dehydration reaction in the presence of strong acids like
Thus, the alkenes formed by the treatment of given alcohol with

Figure 4
The alkenes formed by the treatment of given alcohol with
Want to see more full solutions like this?
Chapter 9 Solutions
Organic Chemistry-Package(Custom)
- 16. Identify each compound as an a cohol, a phenol, or an ether. Classify any alcohols as primary (1"), secondary (2), or tertiary (3"). a. CH,CH,CH,OH CH,CHCH, b. CHO C. CH CHOCH, CH, d.arrow_forwardGive the IUPAC name for each ketone. C-CHCH,CH3 b. a.arrow_forward18. Ketone reduction Dicyclohexyl ketone Reduce the ketone. 1. NaBH4, ethanol 2. H3O+ H OH Dicyclohexylmethanol (88%) (a 2° alcohol)arrow_forward
- What alkenes are formed when each alcohol is dehydrated with TsOH? Label the major product when a mixture results.arrow_forwardGive the IUPAC name for each alcohol но сн CH,CHCCH,CH,CH3 (CH)½CHCH,CHCH,CH3 b. a. CH,CH,CH,OH HO. с. CH,CH,CH,CH, Draw the products formed when each alcohol is dehydrated with H2SO4. Use the Zaitsev rule to predict the major product when a mixture forms. OH он b. -CHCH,CH3 а. Он с. CH3CHCH2CH,CH;CH3arrow_forwardDraw the products of each reaction. CH3 a. CH3-C-CH,CH3 HCI HI OH HBr b. C. OHarrow_forward
- 4. Which compound is an acetal? O. %3D a. I b. II с. III d. IV e. All of thesearrow_forwardList the products of each alcohol reaction. CH3 a. CH,-C-OH CH, NazCrO b. CH3-CH-CH;-CH2-OH c. CH-CH-OH +HCI -arrow_forwardWhich are secondary alcohols? H. CH; CH; c-c-OH H,C H H;C-C CH; H `CHCH, OH II III IV O A. I, II B. II, II C. III, IV O D. I, IVarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





