
Draw the organic product(s) formed when
a.
b.
c.
d.
e.
f.
g.
h.
i.
j.
(a)
Interpretation: The organic product(s) formed by the treatment of
Concept introduction: Alcohols undergo dehydration reaction in the presence of strong acids like
Answer to Problem 9.45P
The organic product(s) formed by the treatment of
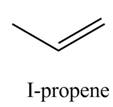
Explanation of Solution
The given reagent is
Alcohols undergo dehydration reaction in the presence of strong acids like
The organic product(s) formed by the treatment of
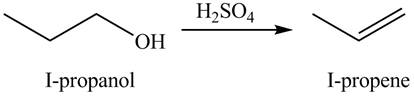
Figure 1
The organic product(s) formed by the treatment of
(b)
Interpretation: The organic product(s) formed by the treatment of
Concept introduction: An alkoxide salt is required to prepare ether. The alkoxide salts are prepared from alcohols through the Bronsted-Lowry acid-base reaction. In this reaction,
Answer to Problem 9.45P
The organic product(s) formed by the treatment of
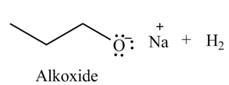
Explanation of Solution
The given reagent is
An alkoxide salt is required to prepare ether. The alkoxide salts are prepared from alcohols through the Bronsted-Lowry acid-base reaction. In this reaction,
The organic product(s) formed by the treatment of
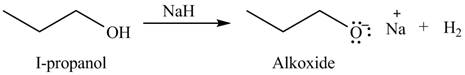
Figure 2
The organic product(s) formed by the treatment of
(c)
Interpretation: The organic product(s) formed by the treatment of
Concept introduction: The reactivity of
Answer to Problem 9.45P
The organic product(s) formed by the treatment of
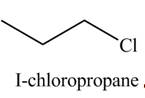
Explanation of Solution
The given reagent is
The reactivity of
The organic product(s) formed by the treatment of
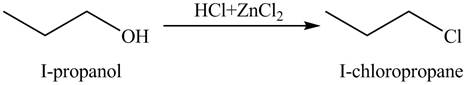
Figure 3
The organic product(s) formed by the treatment of
(d)
Interpretation: The organic product(s) formed by the treatment of
Concept introduction: The reaction of alcohols with halogen acids
Answer to Problem 9.45P
The organic product(s) formed by the treatment of
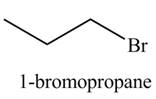
Explanation of Solution
The given reagent is
The reaction of alcohols with halogen acids
The organic product(s) formed by the treatment of
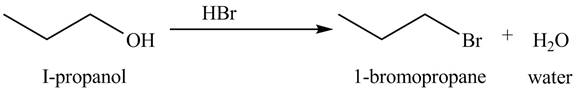
Figure 4
The organic product(s) formed by the treatment of
(e)
Interpretation: The organic product(s) formed by the treatment of
Concept introduction: Alkyl chlorides are obtained by the reaction of
Answer to Problem 9.45P
The organic product(s) formed by the treatment of
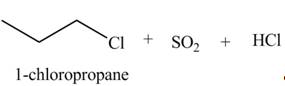
Explanation of Solution
The given reagent is
Alkyl chlorides are obtained by the reaction of
The organic product(s) formed by the treatment of
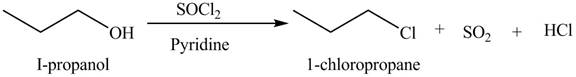
Figure 5
The organic product(s) formed by the treatment of
(f)
Interpretation: The organic product(s) formed by the treatment of
Concept introduction: Alkyl bromides are obtained by the reaction of
Answer to Problem 9.45P
The organic product(s) formed by the treatment of
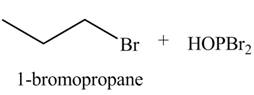
Explanation of Solution
The given reagent is
Alkyl bromides are obtained by the reaction of
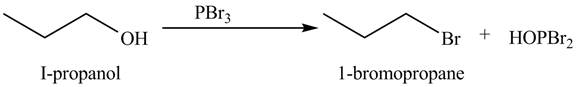
Figure 6
The organic product(s) formed by the treatment of
(g)
Interpretation: The organic product(s) formed by the treatment of
Concept introduction: Alcohols are converted into alkyl tosylates by treatment with
Answer to Problem 9.45P
The organic product(s) formed by the treatment of
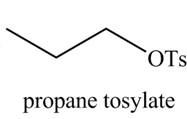
Explanation of Solution
The given reagent is
Alcohols are converted into alkyl tosylates by treatment with
The organic product(s) formed by the treatment of
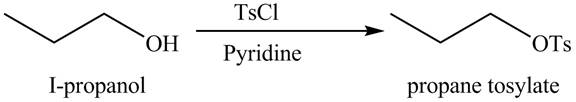
Figure 7
The organic product(s) formed by the treatment of
(h)
Interpretation: The organic product(s) formed by the treatment of
Concept introduction: An alkoxide salt is required to prepare ether. The alkoxide salts are prepared from alcohols through the Bronsted-Lowry acid-base reaction. In this reaction,
The formed alkoxide is allowed to react with an alkyl halide to obtain ether. The mechanism of the reaction is
Answer to Problem 9.45P
The organic product(s) formed by the treatment of
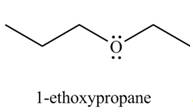
Explanation of Solution
The given reagents are
An alkoxide salt is required to prepare ether. The alkoxide salts are prepared from alcohols through the Bronsted-Lowry acid-base reaction. In this reaction,
The formed alkoxide is allowed to react with an alkyl halide to obtain ether. The mechanism of the reaction is
The organic product(s) formed by the treatment of
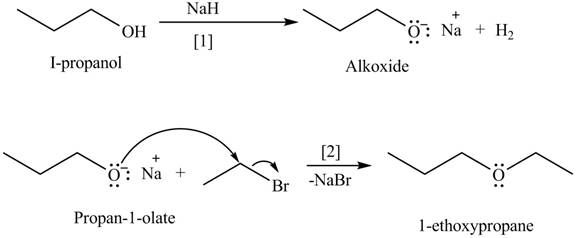
Figure 8
The organic product(s) formed by the treatment of
(i)
Interpretation: The organic product(s) formed by the treatment of
Concept introduction: Alcohols are converted into alkyl tosylates by treatment with
The formed alkyl tosylate reacts with strong nucleophile
Answer to Problem 9.45P
The organic product(s) formed by the treatment of
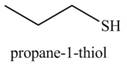
Explanation of Solution
The given reagents are
Alcohols are converted into alkyl tosylates by treatment with
The formed alkyl tosylate react with strong nucleophile
The organic product(s) formed by the treatment of
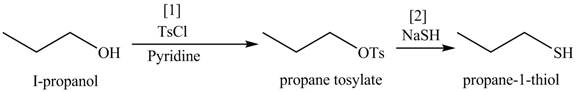
Figure 9
The organic product(s) formed by the treatment of
(j)
Interpretation: The organic product(s) formed by the treatment of
Concept introduction: Alcohols undergo dehydration reaction in the presence of
Answer to Problem 9.45P
The organic product(s) formed by the treatment of
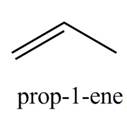
Explanation of Solution
The given reagent is
Alcohols undergo dehydration reaction in the presence of
The organic product(s) formed by the treatment of
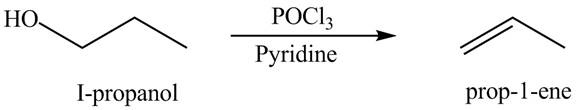
Figure 10
The organic product(s) formed by the treatment of
Want to see more full solutions like this?
Chapter 9 Solutions
ORGANIC CHEMISTRY
Additional Science Textbook Solutions
Organic Chemistry
Chemistry: Atoms First
Chemistry: Structure and Properties (2nd Edition)
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
Introductory Chemistry (5th Edition) (Standalone Book)
- 19. Choose the correct reagents/reactants to synthesize the following compound. 1. NaH 1. NaH A. OH 2. OH 2. Br Br 3. H3O* С. 1. NaH 1. NaH HO 2. HO 2. Br Br 3. H;0* B. D.arrow_forward16. Predict the products of the following reactions. ( a. Br 1. Mg/ether 2. CO2 3. H,O* b. NH d. HO Br CN 1. LDA e. 2. CH;CH,Iarrow_forwardWhat is the product of the following reaction? 1. B2Н6. THF 2. NaOH, H2O2 он HO С. А. В. D.arrow_forward
- What is the major product in the following reaction? -CH, Br2 H,0 CH, OH Br a. CH, он b. CH, OH C. CH; OH d.arrow_forwardWhat is the major product? OH 1. TSCI, pyrndine 2. NaCN CN O b. CN O d . NCarrow_forward4. Which is the major product of this reaction? а. b. C. OH d. 1. FeCl3, CI 2. Hа/Pdarrow_forward
- Choose the correct product for each reaction. A. B. C. D. PPh3 H PPh3 || I ။arrow_forwardWhat is the product of this reaction? HO A. B. O A OB O C OD C OH H* cat. C. D.arrow_forwardQUESTION 2 The following is a proposed synthetic strategy that uses one starting alcohol in two separate single-step reactions (I and II) to make a pair of diene and dienophile which together undergo a Diels-Alder reaction to give a Diels-Alder product. The Diels-Alder product is subsequently converted to the final ether in a three-step synthesis (III, IV and V). Identify the reagent(s) from Table I required for each of those three sysnthese and place the number (A1, A2 reagent(s) in the given box right next to the corresponding step numbers (I to V) of the synthesis. DO NOT fill in any box with more than one number. (Note: This question is different from the previous synthesis question which allows two numbers for each box). You will be graded based on the number in each box. Do not fill in any single box with more than one number. Do not leave any box blank. B1, B2..., or C1, C2 ...) that represents the (I) diene OH (III, IV and V) Diesl-Alder product (II) dienophile Table I Number…arrow_forward
- Reaction Product/s a. C,H1, + O2 b. Butane+ CI, c. Propanearrow_forward13. Give the major organic product for the following reactions. a. NaH a. NaH a. OH d. b. CH3BR b. HO, OH a. NaH a. NaH b. CH3CH,CH,Br HO- Br b. a. NaH OH a. NaH с. CH3OH b. CI f. b. b.arrow_forward1. CH, CH, - CH,-CH,-CH, + HO 2. H₂C-CH=CH₂ + HBr/H₂O₂ 3. H₂C-CH (OH)-CH₂ + H* 4. Nitrobenzene +? ? anilinearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





