
Concept explainers
Interpretation:
The hybrid orbitals of carbon atoms in the given molecules are to be determined.
Concept introduction:
Hybridization is the combining of atomic orbitals to form hybrid orbitals.
To determine hybridization of an atom, first draw the Lewis structure of the molecule.
Find the number of electron domains around an atom so as to get the number of hybrid orbitals used by the atom for bonding.
When atomic orbitals combine, they form equal number of hybrid orbitals.
The s orbital combines with one, two, or three p orbitals to form
Answer to Problem 36QP
Solution:
a)
b)
c)
d)
e)
a)
Explanation of Solution
The Lewis structure of
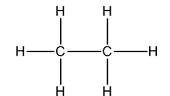
In this, the two carbon atoms are bonded to each other and the three hydrogen atoms by single bonds. Thus, there are four electron domains around each carbon atom. Four electron domainsdepict thepresence of four hybrid orbitals. Thus, in both carbon atoms
b)
The Lewis structure of
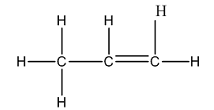
The carbon on the left is bonded to three hydrogen atoms by single bonds and to a carbon atom by a single bond. Thus, there are four electron domains around each carbon atom. Four electron domainsdepict thepresence of four hybrid orbitals. Thus, in this carbon
The central carbon is bonded to one carbon atom by a single bond, to another by double bond and to a hydrogen by a single bond. Thus, there are three electron domains around this carbon atom. Three electron domainsdepict thepresence of three hybrid orbitals. Thus, hybridization of the central carbon is
The carbon on the right is bonded to two hydrogen atoms by a single bond and to a carbon by a double bond. Thus, there are three electron domains around this carbon atom. Three electron domainsdepict thepresence of three hybrid orbitals. Thus, hybridization of the carbon on the right is
Thus, the hybridizations of the carbon atoms, from left to right in the molecule, are
c)
The Lewis structure of
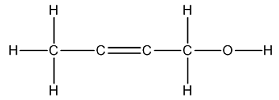
The carbon on left is bonded to three hydrogen atoms by single bonds and to a carbon atom by single bond. Thus, there are four electron domains around each carbon atom. Four electron domainsdepict thepresence of four hybrid orbitals. Thus, in this carbon,
The two carbon atoms in the center are bonded to each other by double bonds and to a carbon by a single bond. Thus, there are two electron domains around each carbon atom. Two electron domains depict the presence of two hybrid orbitals. Thus, in these two carbons, sp hybrid orbitals are present.
The carbon on the right is bonded to two hydrogen atoms by single bonds, to a carbon atom by a single bond and to an oxygen by a single bond. Thus, there are four electron domains around this carbon atom. Four electron domainsdepict the presence of four hybrid orbitals. Thus, in this carbon,
Thus, the hybridizations of the carbon atoms from left to right in the molecule are
d)
The Lewis structure of
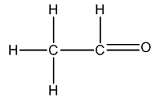
The carbon on the left is bonded to three hydrogen atoms by single bonds and to a carbon atom by a single bond. Thus, there are four electron domains around each carbon atom. Four electron domainsdepict the presence of four hybrid orbitals. Thus, in this carbon
The carbon on the right is bonded to a hydrogen atom by a single bond and to an oxygen atom by a double bond. Thus, there are three electron domains around this carbon atom. Three electron domainsdepict the presence of three hybrid orbitals. Thus, the hybridization of the carbon atom on the right is
Thus, the hybridizations of carbon atoms from left to right in the molecule are
e)
The Lewis structure of
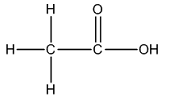
The carbon on left is bonded to three hydrogen atoms by single bonds and to carbon atom by single bond. Thus, there are four electron domains around each carbon atom. Four electron domainsdepict the presence of four hybrid orbitals. Thus, in this carbon
The carbon on the right is bonded to an oxygen atom by a double bond and to another oxygen atom by a single bond. Thus, there are three electron domains around this carbon atom. Three electron domainsdepict the presence of three hybrid orbitals. Thus, the hybridization of the carbonatomon the right is
Thus, the hybridizations of carbon atoms from left to right in the molecule are
Want to see more full solutions like this?
Chapter 9 Solutions
EBK CHEMISTRY
- Aspirin, or acetylsalicylic acid, has the formula C9H8O4 and the skeleton structure (a) Complete the Lewis structure and give the number of bonds and bonds in aspirin. (b) What is the hybridization about the CO2H carbon atom (colored blue)? (c) What is the hybridization about the carbon atom in the benzene-like ring that is bonded to an oxygen atom (colored red)? Also, what is the hybridization of the oxygen atom bonded to this carbon atom?arrow_forwardOctocrylene is an ingredient found in topical sunscreens. It is a water-resistant molecule that helps protect skin against harmful UVA and UVB radiation. Octocrylene Please answer the following questions: (a) What is the hybridisation of each nonhydrogen atom? (b) Are there two unique configurations possible about the C=C double bond? Please explain your answer. (c) Which of the two C-C bonds indicated by the arrows would you expect to be shorter? Please explain your answer.arrow_forward(a) State the hybrid orbitals for C1,C2,O1 and O2 (b) state the number of π bonds present (c) State teh number of C atoms with sp2 hybridization.arrow_forward
- In ozone, O3, the two oxygen atoms on the ends of the molecule are equivalent to one another. (a)What is the best choice of hybridization scheme for the atoms of ozone? (b) For one of the resonance forms of ozone, which of the orbitals are used to make bonds and which are used to hold nonbonding pairs of electrons? (c) Which of the orbitals can be used to delocalize the π electrons? (d) How many electrons are delocalized in the π system of ozone?arrow_forwardThe drawing below shows the overlap of two hybrid orbitalsto form a bond in a hydrocarbon. (a) Which of the followingtypes of bonds is being formed: (i) C¬C σ, (ii) C¬C π,or (iii) C¬H σ? (b) Which of the following could be theidentity of the hydrocarbon: (i) CH4, (ii) C2H6, (iii) C2H4, or(iv) C2H2?arrow_forwardThe structure of caffeine is shown below. (a) Complete the Lewis structure. (b) How many pi bonds are present in caffeine? How many sigma bonds? (c) Identify the hybridization of the carbon atoms. (d) What is the value of the O-C-N angle?arrow_forward
- Convert the following molecular formulas into linebond structures: (a) C3Ha (b) C3H;Br (two possibilities) (c) C3H6 (two possibilities) (d) C2H.O (two possibilities) Identify the bonds in the following molecules as covalent, polar covalent, or ionic (a) BeF2 What is the hybridization of each carbon atom in acetonitrile, CH3C=N? (b) SİH4 (с) СВraarrow_forwardEthyl acetate, C4H8O2, is a fragrant substance used both as asolvent and as an aroma enhancer. Its Lewis structure is (a) What is the hybridization at each of the carbon atomsof the molecule? (b) What is the total number of valenceelectrons in ethyl acetate? (c) How many of the valence electronsare used to make s bonds in the molecule? (d) Howmany valence electrons are used to make p bonds? (e) Howmany valence electrons remain in nonbonding pairs in themolecule?arrow_forwardIn following decomposition reaction:2C3H8(g)⟶ C2H4(g)+ C3H6(g) + CH4(g) + H2(g)For each of the four carbon compounds, do the following: (a) Draw Lewis structure.(b) Predict the geometry about the carbon atom.(c) Determine the hybridization of each type of carbon atom.arrow_forward
- Octocrylene is an ingredient found in topical sunscreens. It is a water-resistant molecule that helps protect skin against harmful UVA and UVB radiation. (a) What is the hybridization of each nonhydrogen atom? (b) Circle all atoms bonded to the acyclic C=C double bond that are required to be in the same plane. (c) Are there two unique configurations possible about the acyclic C=C double bond? Explain. (d) Which of the two C-C single bonds indicated by arrows would you expect to be shorter? Explain. Octocrylenearrow_forwardIn the hydrocarbon: (a) What is the hybridization at each carbon atom in themolecule? (b) How many σ bonds are there in the molecule?(c) How many π bonds? (d) Identify all the 120° bond anglesin the molecule.arrow_forwardWrite the name of the molecular bonding orbital (sigma, sigma star, pí, pí star) that results when the following atomic orbitals overlap. (a) (b) (c) + + ㅇㅇarrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

