
a)

Interpretation:
To predict the product of the reaction shown with regiochemistry, if relevant.
Concept introduction:
Catalytic hydrogenation is a heterogeneous process which occurs with syn stereochemistry. Both hydrogens adsorbed on the catalytic surface add to the double bond from the same face.
To predict:
The product of the reaction shown with regiochemistry, if relevant.
Answer to Problem 42AP
The product of the reaction is ethylbenzene.
Explanation of Solution
The addition of hydrogen takes place to the double bond in the side chain with syn stereochemistry. Both hydrogens add to the double bond from the same face to yield ethylbenzene.
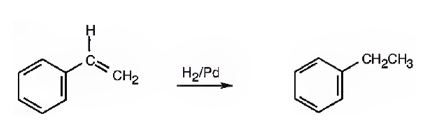
The product of the reaction is ethylbenzene.
b)
Interpretation:
To predict the product of the reaction shown with regiochemistry, if relevant.
Concept introduction:
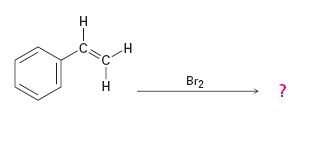
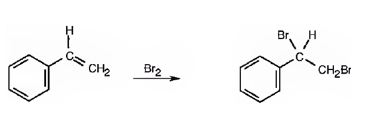
Addition of bromine to the double bond in
To predict:
The product of the reaction shown with regiochemistry, if relevant.
Answer to Problem 42AP
The product of the reaction is
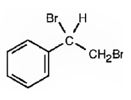
Explanation of Solution
In the first step the addition of bromine to the double bond in 1,2-dimethylcyclohexane results in the formation of a cyclic bromonium ion with the simultaneous elimination of a bromide ion. The large bromonium ion shields one side of the molecule. Hence the attack of the bromide ion occurs from the opposite, unshielded side to yield a trans-product.
The product of the reaction is
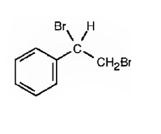
c)
Interpretation:
To predict the product of the reaction shown with regiochemistry, if relevant.
Concept introduction:
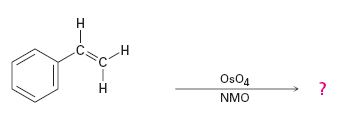
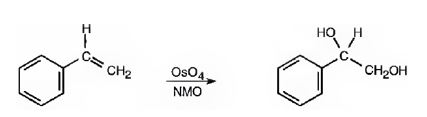
Hydroxylation of double bonds can be carried out directly by treating the alkene with osmium tetroxide, OsO4, in the presence of N-phenylmorpholine N-oxide. The reaction occurs with syn stereochemistry through the formation of a cyclic intermediate, called osmate, formed by the addition of OsO4 to the alkene in a single step. The cyclic osmate is then cleaved to give the cis-1,2-
To predict:
The product of the reaction shown with regiochemistry, if relevant.
Answer to Problem 42AP
The product of the reaction is
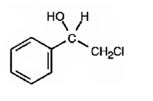
Explanation of Solution
OsO4 adds to the double bond in the side chain from the same side to yield a cyclic osmate in a single step. When treated with NaHSO3 the cyclic osmate gets cleaved to give the diol.
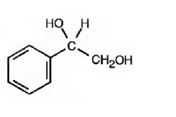
The product of the reaction is
d)
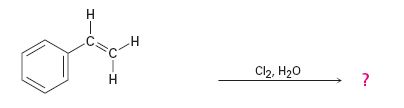
Interpretation:
To predict the product of the reaction shown with regiochemistry, if relevant.
Concept introduction:
The addition of chlorine to alkenes in the presence of water results in the anti addition of the halohydrin, HOCl, to the double bond. In the first step a cyclic chloronium ion is formed by the attack of the π electrons of the double bond on chlorine. In the second step water attacks the chloronium ion from the least shielded side to give an anti addition product. The addition obeys Markovnokov orientation. The negative part (OH) adds to the doubly bonded carbon atom which has more number of substituents.
To predict:
The product of the reaction shown with regiochemistry, if relevant.
Answer to Problem 42AP
The product of the reaction is
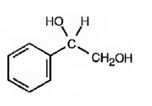
Explanation of Solution
The given alkene has an unsymmetrically substituted double bond in the side chain. In the first step a cyclic chloronium ion is formed by the attack of the double bond on chlorine. In the second step water attacks the the chloronium ion from the opposite less shielded side to give an intermediate which then loses a proton to yield the product.
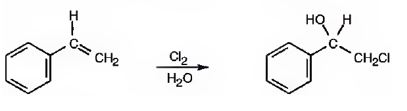
The product of the reaction is
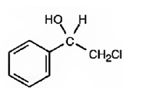
e)
Interpretation:
The product expected in the reaction shown is to be given.
Concept introduction:
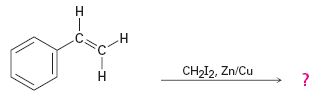
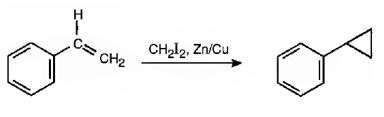
The reaction given is an example of Simmons-Smith reaction. When CH2I2 is treated with Zn/Cu couple, iodomethylzinc iodide, ICH2ZnI, is formed. This ICH2ZnI transfers a CH2 group to the double bond in alkene to form a cyclopropane ring in the product.
To give:
The product expected in the reaction shown.
Answer to Problem 42AP
The product expected in the reaction shown is
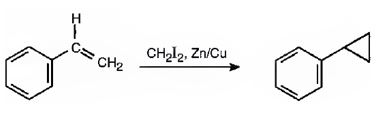
Explanation of Solution
When CH2I2 is treated with Zn/Cu couple, iodomethylzinc iodide, ICH2ZnI, is formed. This ICH2ZnI transfers a CH2 group to the double bond in the side chain to form a cyclopropane ring in the product.
The product expected in the reaction shown is
f)
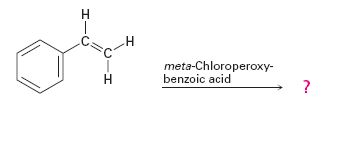
Interpretation:
The product expected in the reaction shown is to be given.
Concept introduction:
Alkenes are oxidized to give
To give:
The product expected in the reaction shown.
Answer to Problem 42AP
The product expected in the reaction shown is
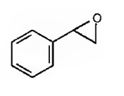
Where RCO3H = meta-chloroperoxybenzoic acid
Explanation of Solution
The oxygen transfer from the peroxy acids to the double bond in the side chain takes place with syn stereochemistry. Both the C-O bonds are formed from the same face of the double bond through a one step process without the formation of an intermediate to yield the product.
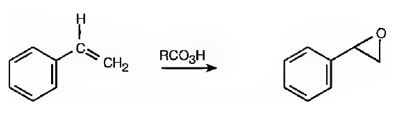
The product expected in the reaction shown is
Want to see more full solutions like this?
Chapter 8 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- Give the structure of the product and/or intermediates of the following reactions. Indicate, whenappropriate, both regiochemistry and stereochemistryarrow_forwardPredict the products and include stereochemistyarrow_forwardprovide the major profucts ( wrote "no reaxtion "if you think so) for the following reactions with correct stereochemistry. Please explain step by step with explanationarrow_forward
- 2) The Diels-Alder reaction, developed by German chemists Otto Diels and Kurt Alder (who received the Nobel Prize in 1950 for their discovery), has great synthetic importance due to the possibility of forming an unsaturated six-membered cycle without involving intermediates ionic. About the reaction, answer: (a) Indicate the reagents necessary for the synthesis of the following compounds, indicating who is the diene and who is the dienophile.arrow_forwardComplete the following reactions, clearly indicating regio-and stereochemistry where applicable. In cases, where ortho-and para-products are formed, draw both.arrow_forwardPredict the products of the following reduction reactions, including stereochemistry where needed. If the reaction product is racemic, indicate that by writing “racemic”. please explain stepsarrow_forward
- Please provide a curved-arrow mechanism for the following reaction. Be sure to use your mechanism and a few words to explain the regiochemistry of the bromines in the final product and why each carbocation intermediate in your mechanism is the preferred intermediate for that step. Include ALL lone pairs and formal charges. Thank you.arrow_forwardtwo positions of anthracene sometimes react more like polyenes thanlike aromatic compounds.(a) Draw a Kekulé structure that shows how the reactive positions of anthracene are the ends ofa diene, appropriate for a Diels–Alder reaction.(b) The Diels–Alder reaction of anthracene with maleic anhydride is a common organic labexperiment. Predict the product of this Diels–Alder reaction.arrow_forwardPredict the major products of the following reactions. No reaction is a possible answer.arrow_forward
- Rank the basicity of the following sets of compounds. And give explanation.arrow_forwardChemistry 3. Complete the following reaction scheme. Give all product(s) and indicate major or minor and any relevant stereochemistry. (а) NaOEt heat (b) heat (c) Br NaCN DMF (d) (e) OH H,SO,/H,PO, heat (f) HBr (g) 1) BH. THE 2) H,0, OH (h) 1) Hg(OAC), H-0 2) NaBH, (i) KMNO, NaOH Coldarrow_forwardBoth pyrrole and pyridine are nitrogen containing heterocycles that are 6π Hückel aromatic. Describe the aromaticity and observed basicity for that molecule, which is more basic, explain and show the acid -base reaction involved.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
