
Concept explainers
(a)
Interpretation: The product of the given
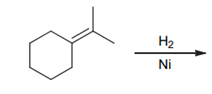
Concept introduction:
Some examples of addition reactions of
(b)
Interpretation: The product of the given chemical reaction is to be interpreted.
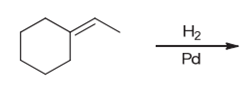
Concept introduction:
Some examples of addition reactions of alkenes are hydrogenation, acid-catalyzed hydration, hydroboration-oxidation, oxymercuration-demercuration, etc. Hydrogenation of alkene leads to the formation of saturated hydrocarbons; alkanes. The addition of hydrogen can occur in the presence of metal catalysts like Raney
(c)
Interpretation: The product of the given chemical reaction is to be interpreted.
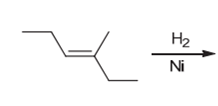
Concept introduction:
Some examples of addition reactions of alkenes are hydrogenation, acid-catalyzed hydration, hydroboration-oxidation, oxymercuration-demercuration, etc. Hydrogenation of alkene leads to the formation of saturated hydrocarbons; alkanes. The addition of hydrogen can occur in the presence of metal catalysts like Raney
(d)
Interpretation: The product of the given chemical reaction is to be interpreted.
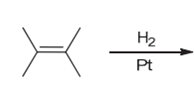
Concept introduction:
Some examples of addition reactions of alkenes are hydrogenation, acid-catalyzed hydration, hydroboration-oxidation, oxymercuration-demercuration, etc. Hydrogenation of alkene leads to the formation of saturated hydrocarbons; alkanes. The addition of hydrogen can occur in the presence of metal catalysts like Raney
(e)
Interpretation: The product of the given chemical reaction is to be interpreted.
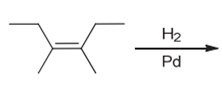
Concept introduction:
Some examples of addition reactions of alkenes are hydrogenation, acid-catalyzed hydration, hydroboration-oxidation, oxymercuration-demercuration, etc. Hydrogenation of alkene leads to the formation of saturated hydrocarbons; alkanes. The addition of hydrogen can occur in the presence of metal catalysts like Raney
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
EBK ORGANIC CHEMISTRY, ENHANCED ETEXT
- Identify the reagents necessary to accomplish each of the following transformations.arrow_forwardWhich of the ketones below could not be prepared by an acid-catalyzed hydration of an alkyne?arrow_forwardIdentify the major and minor product(s) of the following reaction: of Br NaOEt ? ELOHarrow_forward
- Give the product(s) for the following reaction, predicting the major product, anc indicating by which mechanism each is formed. Br MeOH heatarrow_forwardPredict the major product for the following reaction and in the box provided, write thecorresponding compound letter (A, B, C, or D) or write NR if no reaction is expected to occur.arrow_forwardPredict the major products of the following reactions. If it is possible, write all stereoisomers.arrow_forward
- The hemiacetal reaction is also reversible and can also be catalyzed by either acid or base. Hemiacetals are not usually isolated, except in the formation of 5- and 6-membered rings, as often seen in carbohydrate chemistry. Identify the hemiacetal formed from the intramolecular cyclization of the molecule shown. OH OH NaOH ? H CH₂OH OH OH The hemiacetal product is: HOH₂C HO O HOH₂C H OH OH HO OH HO OH HO OH O HOH₂C О OH OH H HO- OH HO OH CH₂OH OH OHarrow_forwardPredict the major product of the following reaction and draw the resonance structure(s) responsible for the regiochemistry of the reaction. HNO3 ? H2SO4arrow_forwardWhich of the following is a major product of the reaction shown below? он NaBH, (еxcess) OCH3 ELOH (A) (B) OH OCH3 но HO (C) (D) O Compound D Compound C OCompound A Compound Barrow_forward
- Predict the product (including stereochemistry) when the following substance reacts under thermal conditions. H H3C- H3C- H.arrow_forwardPredict the major products of the following reaction. If it is possible, write all stereoisomers.arrow_forwardProvide the major organic product of the reaction below explain the mechanismarrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

