
Concept explainers
Interpretation:
- The products of the given reaction have to be predicted.
Concept Introduction:
Resonance Contributor: The appropriate structure with the localized electrons is called a resonance contributor, a resonance structure, or a contributing resonance structure.
Delocalized electrons: The sharing of electrons between two or more atoms known as delocalization of electrons. In order to have delocalized electrons, the system must be planar and have alternative double bonds and single bonds.
Resonance hybrid: The actual structure with delocalized electrons is called a resonance hybrid.
Answer to Problem 57P
Correct Answer:
The compounds A, B, C, E, F, L, M, N, and O have delocalized electrons.
Explanation of Solution
Reason for correct options:
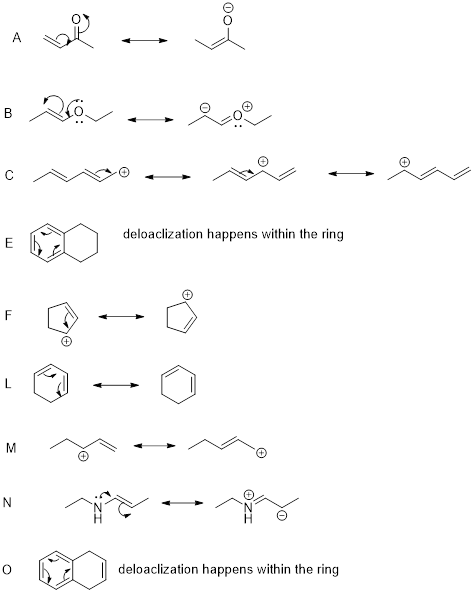
The above compounds all have delocalized electrons, which undergo electron movement. The resonance structures can be drawn as shown above.
Hence, the correct options are A, B, C, E, F, L, M, N, and O.
Reason for in-correct options:
The remaining compounds do not contain delocalized electrons, conjugated system is absent.
Hence, the in-correct options are D, G, H, I, J, and K.
- The compounds containing delocalized electrons were predicted.
Want to see more full solutions like this?
Chapter 8 Solutions
Organic Chemistry; Modified MasteringChemistry with Pearson eText -- ValuePack Access Card; Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
- Explain why alkene A is more stable than alkene B, even though Bcontains more carbon atoms bonded to the double bond. Would youexpect C to be more or less stable than A and B?arrow_forwardThe purine heterocycle occurs commonly in the structure of DNA. a. How is each N atom hybridized? b. In what type of orbital does each lone pair on a N atom reside? c. How many a electrons does purine contain? d. Why is purine aromatic? purinearrow_forwardExplanation for part C and D.arrow_forward
- The following compound is? a. none of the other answers O b. anti-aromatic Oc non-aromatic O d. aromaticarrow_forwardThe purine heterocycle occurs commonly in the structure of DNA.a.How is each N atom hybridized? b.In what type of orbital does each lone pair on a N atom reside? c.How many π electrons does purine contain? d.Why is purine aromatic?arrow_forwardPlease explain A,B, and C. Thank you!arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

