
Concept explainers
(a)
Interpretation: The equation for the reaction that takes place to increase the solubility of
Concept Introduction: The solubility of a salt is defined as amount of salt dissolved in per 100 mL of the solvent at a constant temperature.
(a)
Explanation of Solution
The reaction of aluminium ions and hydroxide ion forms aluminium hydroxide but it can also form
The precipitate is more soluble in very acidic and basic solutions.
The equations for reaction to increase the solubility of
The reaction in acidic solution is represented as follows:
The reaction in basic medium is represented as follows:
(b)
Interpretation: This is to be shown that the solubility of
Concept Introduction:
For a salt, the expression for solubility product can be written as follows:
The solubility product constant can be represented as follows:
(b)
Explanation of Solution
The relation between K,
Or,
The equilibrium constant expression can be represented as follows:
For the above reaction,
If ionization constant of water is used in this expression, the above expression can be rearranged as follows:
On rearranging,
Or,
The solubility product of
This can be rearranged as follows:
Also,
From equation (1) and (2),
(b)
Interpretation: The solubility of
Concept Introduction: The relation between solubility and hydrogen ion concentration can be represented as follows:
The relation between pH and hydrogen ion concentration can be represented as follows:
(b)
Explanation of Solution
The given value of K is 40.0 and solubility product constant is
The solubility of
Putting the value of K,
Or,
Now, for pH value 4, the hydrogen ion concentration will be
Similarly, for pH value 5, the hydrogen ion concentration will be
Thus, for pH value 6 -12, the solubility of
| pH | |
| 6 | |
| 7 | |
| 8 | |
| 9 | |
| 10 | |
| 11 | |
| 12 | |
| 13 | |
| 14 |
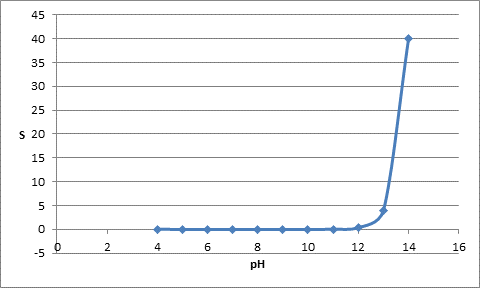
The table for pH and solubility can be represented as follows:
| pH | S |
| 4 | 0.02 |
| 5 | |
| 6 | |
| 7 | |
| 8 | |
| 9 | |
| 10 | |
| 11 | |
| 12 | 0.4 |
| 13 | 4 |
| 14 | 40 |
The plot can be represented as follows:
Want to see more full solutions like this?
Chapter 8 Solutions
Chemical Principles
- How would the solubility of calcium fluoride be affected by the presence of fluoride ion from another source? What is the solubility of calcium fluoride in a saturated solution of barium fluoride? How does this compare with the value of the solubility of calcium fluoride found in Example 17.4? Is this what you expect?arrow_forwardSome barium chloride is added to a solution that contains both K2SO4 (0.050 M) and Na3PO4 (0.020 M). (a) Which begins to precipitate first: the barium sulfate or the barium phosphate? (b) The concentration of the first anion species to precipitate, either the sulfate or phosphate, decreases as the precipitate forms. What is the concentration of the first species when the second begins to precipitate?arrow_forwardSolubility is an equilibrium position, whereas Ksp is an equilibrium constant. Explain the difference.arrow_forward
- A solution contains 0.00740 M calcium ion. A concentrated sodium fluoride solution is added dropwise to precipitate calcium fluoride (assume no volume change). a At what concentration of F does precipitate start to form? b When [F] = 9.5 104 M, what is the calcium-ion concentration? What percentage of the calcium ion has precipitated?arrow_forwardAlthough silver chloride is insoluble in water, it readily dissolves upon the addition of ammonia. AgCl(s)+2NH3(aq)Ag(NH3)2+(aq)+Cl(aq) a What is the equilibrium constant for this dissolving process? b Ammonia is added to a solution containing excess AgCl(s). The final volume is 1.00 L and the resulting equilibrium concentration of NH3 is 0.80 M. Calculate the number of moles of AgCl dissolved, the molar concentration of Ag(NH3)2+, and the number of moles of NH3 added to the original solution.arrow_forwardAn analytical chemist has a solution containing chloride ion, Cl. She decides to determine the amount of chloride ion in the solution by titrating 50.0 mL of this solution by 0.100 M AgNO3. As a way to indicate the endpoint of the titration, she added 1.00 g of potassium chromate, K2CrO4 (see Figure 17.5). As she slowly added the silver nitrate to the solution, a white precipitate formed. She continued the titration, with more white precipitate forming. Finally, the solution turned red, from another precipitate. The volume of the solution at this point was 60.3 mL. How many moles of chloride ion were there in the original solution? How many moles of chloride ion were there in the final solution? You may make any reasonable approximations.arrow_forward
- Sufficient sodium cyanide, NaCN, was added to 0.015 M silver nitrate, AgNO3, to give a solution that was initially 0.108 M in cyanide ion, CN. What is the concentration of silver ion, Ag+, in this solution after Ag(CN)2 forms? The formation constant Kf for the complex ion Ag(CN)2 is 5.6 1018.arrow_forwardHydrogen iodide gas decomposes to hydrogen gas and iodine gas: 2HI(g)H2(g)+I2(g)To determine the equilibrium constant of the system, identical one-liter glass bulbs are filled with 3.20 g of HI and maintained at a certain temperature. Each bulb is periodically opened and analyzed for iodine formation by titration with sodium thiosulfate, Na2S2O3. I2(aq)+2S2O32(aq)S4O62(aq)+2 I(aq)It is determined that when equilibrium is reached, 37.0 mL of 0.200 M Na2S2O3 is required to titrate the iodine. What is K at the temperature of the experiment?arrow_forwardThe solubility of silver sulfate, Ag2SO4, in water has been determined to be 8.0 g/L. What is the solubility in 0.45 M sodium sulfate, Na2SO4?arrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





