
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
5th Edition
ISBN: 9780321967466
Author: Karen C. Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 16CI
The compound butyric acid gives rancid butter its characteristics door. (6.7, 7.4,7.5)
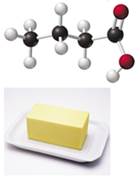
- If black spheres are carbon atoms, white spheres are hydrogen atoms, and red spheres are oxygen atoms, what is the molecular formula of butyric acid?
- What is the molar mass of butyric acid?
- How many grams of butyric acid contain 3.28 × 1023 atoms of oxygen?
- How many grams of carbon are in 5.28 g of butyric acid?
- Butyric acid has a density of 0.959 g/mL at 20?. How many moles of butyric acid are contained in 1.56 mL of butyric acid?
- Identify the bonds C-C, C-H and C-O in a molecule of butyric acid as polar covalent or nonpolar covalent.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
6. (3 points)
Hydralazine is a drug used to treat heart disease. It is 59.99% carbon, 5.03% hydrogen, and 34.98% nitrogen
and has a molecular mass of 160.178 g/mol. What is the molecular formula for hydralazine?
(3.10)A compound has a molar mass of 126.12 g/mol and its empirical formula is C₂H₂O.
Find the molecular formula and fill in each blank with a whole number.
H
< Previous
(5.5, 5.7 & 5.8) Classify the following reaction.
Ba(NO3)2(aq) + K₂SO4(aq) -->
O acid-base reaction
O gas-forming reaction
O decomposition reaction
O precipitation reaction
Chapter 8 Solutions
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Ch. 8.1 - Prob. 8.1QAPCh. 8.1 - Prob. 8.2QAPCh. 8.1 - Prob. 8.3QAPCh. 8.1 - Prob. 8.4QAPCh. 8.1 - Prob. 8.5QAPCh. 8.1 - Prob. 8.6QAPCh. 8.1 - Prob. 8.7QAPCh. 8.1 - Prob. 8.8QAPCh. 8.2 - Why do scuba divers need to exhale air when they...Ch. 8.2 - Why does a sealed bag of chips expand when you...
Ch. 8.2 - The air in a cylinder with a piston has a volume...Ch. 8.2 - Prob. 8.12QAPCh. 8.2 - Prob. 8.13QAPCh. 8.2 - Prob. 8.14QAPCh. 8.2 - Prob. 8.15QAPCh. 8.2 - Prob. 8.16QAPCh. 8.2 - Prob. 8.17QAPCh. 8.2 - Prob. 8.18QAPCh. 8.2 - Prob. 8.19QAPCh. 8.2 - Prob. 8.20QAPCh. 8.2 - Prob. 8.21QAPCh. 8.2 - Prob. 8.22QAPCh. 8.3 - Prob. 8.23QAPCh. 8.3 - Prob. 8.24QAPCh. 8.3 - Prob. 8.25QAPCh. 8.3 - Prob. 8.26QAPCh. 8.3 - Prob. 8.27QAPCh. 8.3 - Prob. 8.28QAPCh. 8.4 - Prob. 8.29QAPCh. 8.4 - Prob. 8.30QAPCh. 8.4 - Prob. 8.31QAPCh. 8.4 - Prob. 8.32QAPCh. 8.4 - Prob. 8.33QAPCh. 8.4 - Prob. 8.34QAPCh. 8.5 - Prob. 8.35QAPCh. 8.5 - Prob. 8.36QAPCh. 8.5 - Prob. 8.37QAPCh. 8.5 - Prob. 8.38QAPCh. 8.6 - Prob. 8.39QAPCh. 8.6 - Prob. 8.40QAPCh. 8.6 - Prob. 8.41QAPCh. 8.6 - Prob. 8.42QAPCh. 8.6 - Prob. 8.43QAPCh. 8.6 - Prob. 8.44QAPCh. 8.6 - Prob. 8.45QAPCh. 8.6 - Prob. 8.46QAPCh. 8.7 - Prob. 8.47QAPCh. 8.7 - Prob. 8.48QAPCh. 8.7 - Prob. 8.49QAPCh. 8.7 - Prob. 8.50QAPCh. 8.7 - Prob. 8.51QAPCh. 8.7 - Prob. 8.52QAPCh. 8.7 - Prob. 8.53QAPCh. 8.7 - Prob. 8.54QAPCh. 8.7 - Prob. 8.55QAPCh. 8.7 - Prob. 8.56QAPCh. 8.7 - Prob. 8.57QAPCh. 8.7 - Prob. 8.58QAPCh. 8.7 - Prob. 8.59QAPCh. 8.7 - Prob. 8.60QAPCh. 8.8 - Prob. 8.61QAPCh. 8.8 - Prob. 8.62QAPCh. 8.8 - Prob. 8.63QAPCh. 8.8 - Prob. 8.64QAPCh. 8.8 - Prob. 8.65QAPCh. 8.8 - Prob. 8.66QAPCh. 8.8 - Prob. 8.67QAPCh. 8.8 - Prob. 8.68QAPCh. 8.8 - Prob. 8.69QAPCh. 8.8 - Prob. 8.70QAPCh. 8 - Prob. 8.71UTCCh. 8 - Prob. 8.72UTCCh. 8 - Prob. 8.73UTCCh. 8 - Prob. 8.74UTCCh. 8 - Prob. 8.75UTCCh. 8 - Prob. 8.76UTCCh. 8 - Prob. 8.77UTCCh. 8 - Prob. 8.78UTCCh. 8 - Prob. 8.79AQAPCh. 8 - Prob. 8.80AQAPCh. 8 - Prob. 8.81AQAPCh. 8 - Prob. 8.82AQAPCh. 8 - Prob. 8.83AQAPCh. 8 - Prob. 8.84AQAPCh. 8 - Prob. 8.85AQAPCh. 8 - Prob. 8.86AQAPCh. 8 - Prob. 8.87AQAPCh. 8 - Prob. 8.88AQAPCh. 8 - Prob. 8.89AQAPCh. 8 - Prob. 8.90AQAPCh. 8 - Prob. 8.91AQAPCh. 8 - Prob. 8.92AQAPCh. 8 - Prob. 8.93CQCh. 8 - Prob. 8.94CQCh. 8 - Prob. 8.95CQCh. 8 - Prob. 8.96CQCh. 8 - Prob. 8.97CQCh. 8 - Prob. 8.98CQCh. 8 - Prob. 8.99CQCh. 8 - Prob. 8.100CQCh. 8 - Prob. 8.101CQCh. 8 - Prob. 8.102CQCh. 8 - Prob. 8.103CQCh. 8 - Prob. 8.104CQCh. 8 - Prob. 13CICh. 8 - Prob. 14CICh. 8 - Prob. 15CICh. 8 - The compound butyric acid gives rancid butter its...Ch. 8 - Prob. 17CICh. 8 - Automobile exhaust is a major cause of air...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- (3.9) How many grams of hydrogen are present in 255 mL of propane at room temperature? The density of propane is 0.493 g/mL at room temperature. O 45.6 g O 11.4 g O 22.8 g O 5.70 garrow_forward(3.7)Classify each of the following as an atomic element, molecular element, molecular compound, or ionic compound. tin(IV) phosphide [Choose ] gold [Choose] carbon monoxide [Choose] V bromine [Choose ] < Previousarrow_forward(5.8)Which of the following reactions will form a gaseous product? O H₂CO3(aq) + Pb(NO3)2(aq) O NaOH(aq) + HNO3(aq) O None of these O Na₂SO3(aq) + H₂SO4(aq) ◄ Previousarrow_forward
- 20 cm3 of hydrogen peroxide solution of unknown concentration reacts completely with 12.15 cm3 0.04 mol/dm3 of potassium permanganate solution in sulfuric acid media. Calculate the mass concentration of hydrogen peroxide in g/dm3. How many cm3 (USC) of oxygen is formed? (2.07; 29.77)arrow_forward8. 9. 10. 4 Fe + 3 0₂ → 2 Fe₂O3 If you have 43 grams of oxygen, how many mols of iron(III) oxide will be formed? If you have 7.5 mol of iron, how many mols of oxygen would be required? If you have 125 g of iron, how many grams of iron(III) oxide will be formed? was per the an de and the red nox 91arrow_forward(3.5)Which of the following compounds contains the metal ion with roman numeral (II) in its name? O CUCO3 ZnCl₂ O Pb(SO4)2 O Hg₂Cl₂arrow_forward
- (5.2, Similar to For More Practice 5.1) What mass of NaOH (in grams) do you need to make 250.0 mL of a 1.50 M NaOH solution? O 6.67 g O 22.3 g O 44.6 g O 15.0 garrow_forward(Q28) How many grams of phosphine (PH3) gas can form when an excess of solid phosphorus (P4) reacts in a sealed chamber with 56.2 liters of hydrogen (H2) gas at STP? (3 sf)arrow_forward6.(0-2) Black powder was invented in China in the 9th century. It was used as an explosive with propelling properties. Today it is used in fireworks, model rocket engines, and replica black powder weapons. Black powder consists of potassium nitrate (potassium nitrate, KN03), charcoal and sulfur. A variety of chemical reactions take place when this mixture is exploded. One of these is shown below. 2KNO, +S+ 30 → K, S+N, ↑+3C0, 1 Knowing that carbon makes up 15% of the black powder, calculate the total volume of gases (under normal conditions) released when 0.1 kg of black powder explodes. Assume that only the chemical reaction given above occurs.arrow_forward
- A student sets up and solves the following equation to solve a problem in solution stoichiometry. Fill in the missing part of the student's equation. (47.6) 65.08 mol 1 mL -3 10 L mol (3²) 3.2 L = 2.3 x 10 mL x10 ロ・ロ X μarrow_forwardHow many different chemical compounds are in the gas phase? 2. 3. 4.arrow_forwardThe decomposition of solid aluminum oxide results in the production of solid aluminum and molecular oxygen gas. If 0.443 moles of aluminum oxide decompose, how many moles of molecular oxygen are generated? (3 sf)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY