
Concept explainers
a)
Interpretation:
To name the
Concept introduction:
The longest carbon chain containing the double bond to be chosen. Based on the name of the parent compound - the alkene name ends with the suffix -ene. The chain is to be numbered from the end that gives the lowest number to the carbon in double bond. Substituents are to be numbered according to their positions in the chain and listed alphabetically. The position of the double bond is indicated by giving the number of the first alkene carbon before the name of the parent name.
In skeletal structures the carbon and hydrogen atoms are not usually shown. Instead carbon atoms are assumed to be at each intersection of two lines and at the end of each line. Hydrogen atoms required are fitted mentally having in mind the valence of carbon is four. Atoms other than carbon and hydrogen are shown.
To name:
The alkene given and to draw its skeletal structure.
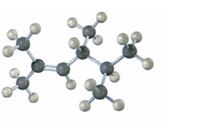
Answer to Problem 22VC
The IUPAC name of the compound is 2, 4, 5-trimethyl-2-hexene. Its skeletal structure is,
Explanation of Solution
The compound given has a continuous chain of six carbons with a double bond between C2 and C3 and three methyl substituents on C2, C4 and C5. Hence the name 2, 4, 5-trimethyl-2-hexene.
The IUPAC name of the compound is 2, 4, 5-trimethyl-2-hexene. Its skeletal structure is,

b)
Interpretation:
To name the alkene given and to draw its skeletal structure.
Concept introduction:
The maximum number of carbons in the ring is counted. Based on the name of the parent cycloalkane - the cycloalkene is named with the suffix -ene. The cycloalkane is numbered such that the double bond is in between C1 & C2 and the first substituent has the lowest number possible. Usually the position of a double bond is not shown in the name because it is always between C1 & C2. In dienes and trienes, however the position of double bonds is shown.
In skeletal structures the carbon and hydrogen atoms are not usually shown. Instead carbon atoms are assumed to be at each intersection of two lines and at the end of each line. Hydrogen atoms required are fitted mentally having in mind the valence of carbon is four. Atoms other than carbon and hydrogen are shown.
To name:
The alkene given and to draw its skeletal structure.
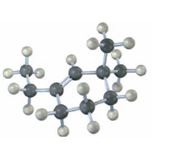
Answer to Problem 22VC
The IUPAC name of the compound is 1-ethyl-3, 3-dimethylcyclohexene. Its skeletal structure is,
Explanation of Solution
The compound given has a six membered ring with an ethyl group on C1 and two methyl groups both on C3. Hence the name is 1-ethyl-3, 3-dimethylcyclohexene.
The IUPAC name of the compound is 1-ethyl-3, 3-dimethylcyclohexene. Its skeletal structure is,

Want to see more full solutions like this?
Chapter 7 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- What is the name of the organic reactant and the resulting product of the reaction below? H H H H H H H-C-C-ċ=ċ-ċ-c-H + CI-CI H H H H O reactant: cis-3-hexene; product: 3,4-dichlorohexane reactant: trans-3-hexene; product: 3,4-dichlorohexane O reactant: cis-3-hexene product: 3,4-chlorohexane O reactant: cis-3-hexene product: dichlorohexanearrow_forwardAn alkene with the molecular formula C10H18is treated with ozone and then with zinc and acetic acid. The only product isolated from these reactions is: What is the structure of the alkene? I O III OV O IV ΟΙ IV II Varrow_forwardDraw the structure of an alkane with molecular formula C7H16 that contains (a) one 4° carbon; (b) only 1° and 2° carbons; (c) 1°, 2°, and 3° hydrogens.arrow_forward
- 5 Write the molecular formula for each alkane. Y \ y t (a) (c) HE8 iw 146 (b)- eriam in Beris mal bond HO H ox dT O HOH w ar HO HO (s) HOarrow_forwardDraw the constitutional isomer formed when the following alkenes are treated with each set of reagents: [1] H2O, H2SO4; or [2] BH3 followed by H2O2, −OH.arrow_forwardIV. Compounds A and B are isomers of the molecular formula C9H19Br. Both yield the same alkene Cin an elimination reaction. Hydrogenation of C yields the product 2,3,3,4 tetramethyl pentane. What are the structures of A, B, and C?arrow_forward
- Give the proper IUPAC name for the following compounds: CH,- CH, CH,- CH - CH -CH, CH, Br CH CH, Br H:-CH; CH - CH-CH, H;C CH, CH, CH, CH- CH- CH, c=C H,C-CH, CH, CH- CH, H H ОН CIarrow_forward7.33 a-Farnesene is a constituent of the natural wax found on apples. What is its IUPAC name, including stereochemistry? 7.34 Menthene, a hydrocarbon found in mint plants, has the systematic name 1-isopropyl-4-methylcyclohexene. Draw its structure. 7.35 Draw and name the six alkene isomers, C5H1o, including E,Z isomers. a-Famesene 8.22 Name the following alkenes, and predict the products of their reaction with (1) meta-chloroperoxybenzoic acid, (2) KMnO4 in aqueous acid, and (3) 03, followed by Zn in acetic acid: (a) (b) (a) 8.23 Draw the structures of alkenes that would yield the following alcohols on hydration (red = O). Tell in each case whether you would use hydroboration- oxidation or oxymercuration-demercuration. (b)arrow_forwardStructure A Cis Isomer H₂C Trans Isomer Name of the Alkene H C=C CH₂CH₂ H [Choose ] [Choose ] Pentene Propene Hexene Structure A Structure B [Choose ] H₂C H C=C Structure B I CH₂CH₂arrow_forward
- 5B In the following reactions, mixtures of alkenes and ethyl ethers are formed. Draw their structures. Explain which is or are likely to be the main product(s) in each reaction. In case of formation of two isomers of alkenes, explain which is formed in greater proportion CH3 CH3 H3C-C H -Br CH3 EtOHarrow_forwardWhat alkene is the major productwhen 2-bromo-2-methylpentane is treated with sodium ethoxide in ethanol?arrow_forward4-Give the IUPAC names of the following compounds: (a) CH,CH₂C=CCOOH (d) CH, COOH (b) CH₂CH(NH₂)CH(OH)COOH NO₂ O.N COOH NO₂ (c) (CH;),C=CHCOOH (1) OHarrow_forward
