
Concept explainers
(a)
Interpretation:
The difficulty of that would come on models of the cis and trans isomers of
Concept introduction:
The organic compounds which consist of two cyclic rings fused together are known as bicyclic compounds. Fused rings, bridged rings, and spiro rings are the examples of bicyclic compounds. The stability of bicyclic compounds depends on the ring strain.
Answer to Problem 7.21P
The cis isomers of
Explanation of Solution
The structure of cis and trans isomers of
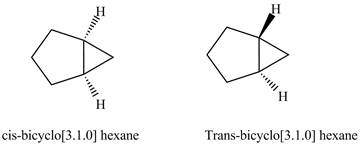
Figure 1
The formation of trans isomers of
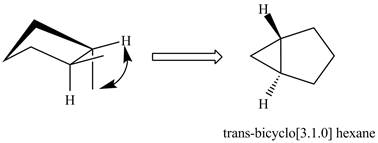
Figure 2
In the formation of trans isomers of
The formation of cis isomers of
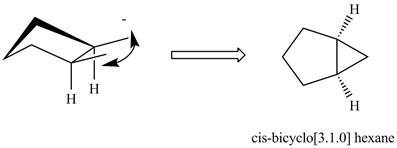
Figure 3
In the formation of cis isomers of
It is easier to form a three-membered ring by the methyl at the cis position and the methyl groups at trans position. Therefore, cis isomers of
(b)
Interpretation:
The difficulty of that would come on models of
Concept introduction:
The organic compounds which consist of two cyclic rings fused together are known as bicyclic compounds. Fused rings, bridged rings and spiro rings are the examples of bicyclic compounds. The stability of bicyclic compounds depends on the ring strain.
Answer to Problem 7.21P
The compound
Explanation of Solution
The structure of
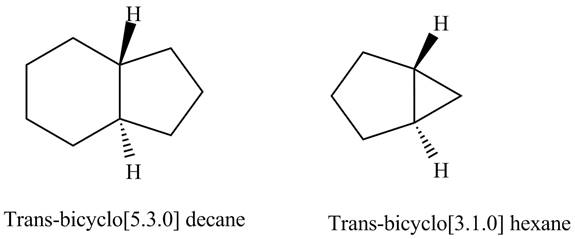
Figure 4
The formation of
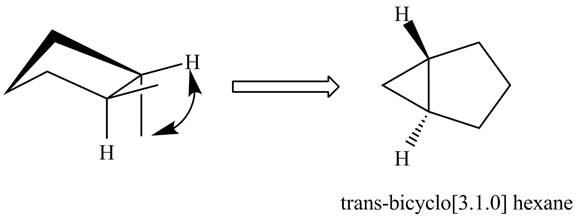
Figure 2
In the formation of
The formation of
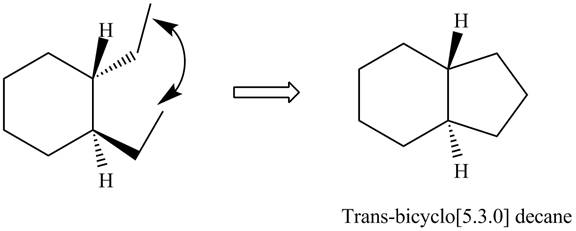
Figure 5
In the formation of
The methyl groups in case of
Want to see more full solutions like this?
Chapter 7 Solutions
EBK ORGANIC CHEMISTRY
- Draw the structure of cis-1,2-dimethylcyclohexane. • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • You do not have to explicitly draw H atoms.arrow_forward4.(a). Draw the structures of (i) cis-1,4-Dibromocyclohexane and (ii) trans-1,4- Dibromocyclohexane. 4(b). Draw the most stable conformers of (i) cis-1,4-Dibromocyclohexane and (ii) trans-1,4-Dibromocyclohexane. 4(c)Which is more stable? (i) cis-1,4-Dibromocyclohexane or (ii) trans-1,4- Dibromolcyclohexane. Explain the reason for your choice.arrow_forwardDraw the structure(s) of the cycloalkanes with the chemical formula C7H₁4 that DO NOT exhibit cis-trans isomerism. The ring must contain 5 or more carbons. • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. • Separate structures with + signs from the drop-down menu. / Sn [Farrow_forward
- What is the structure of bicyclo[1.1.0]butan-2-amine?arrow_forwardSolve AH for each (i) C₂H(g)+O₂(g) (ii) CH(g)+Cl/g) →—> CO₂(g)+H₂O(g), unbalanced —> CCL(1)+HCl(g), unbalancedarrow_forwardCalculate the energy needed (kJ/mol) for the least and the most stable chair conformers of the molecule below. [Energy (kJ/mol): 1,3-diaxial (H- CH3) strain= 3.8; 1,3-diaxial (H-OH) strain= 2.0; 1,3-diaxial (CH3-OH) strain=18.6.]. Write your answer in format--> the least stable:_ kJ/mol; the most stable: kJ/mol * OH H;Carrow_forward
- Calculate strain energy for the conformer pictured below, using strain energy increments from the table. Strain Energy for Alkanes Interaction / Compound kJ/mol H: H eclipsing 4.0 H: CH3 eclipsing CH3 CH3 eclipsing gauche butane H Strain energy = cyclopropane cyclobutane cyclopentane cycloheptane cyclooctane (Calculate your answer to the nearest 0.1 energy unit, and be sure to specify units, kJ/mol or kcal/mol. The answer is case sensitive.) H CH3 ball & stick V + labels 5.8 11.0 3.8 115 H kcal/mol 110 26.0 26.2 40.5 1.0 1.4 2.6 0.9 27.5 26.3 6.2 6.3 9.7arrow_forward+ Br₂ CH₂Cl₂ You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. Do not include counter-ions, e.g., Na, I", in your answer.arrow_forward(a) (b) 1. CH3MgBr 2. H3O+ H3C OH I H3C-C-C-CH3 H3C H ОН H CH3 CH3 H3C CH3 H3C H₂SO4 C=Carrow_forward
- 1. Which of the following formulae represent the same compound? (1) CH;CH,CH;CHCH3 (3) CH-CНCH, 1. CH2 (2) CH3 CH3 CHCH;CH2 CH3 CH3 CH;CH3 (4) CH;CHCH,CH,CH3 (5) CH;CH,CHCH;CH3 CH3 CH3 A. (2) and (3) only В. (1), (2) and (4) only С. (1), (4) and (5) only D. (1), (2), (3) and (4) onlyarrow_forwardConsider 1,2-dimethylcyclohexane.Q: Which isomer, cis or trans, is more stable and why?arrow_forwardDraw the structure(s) of all of the possible monochloro derivatives of 2,2-dimethylpropane, C3H11CI. • You do not have to consider stereochemistry. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right co Separate structures with + signs from the drop-down menu. opy aste ChemDoodle >arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

