
Chemistry: Atoms First (Comp. Instructor's)
3rd Edition
ISBN: 9781259923081
Author: Burdge
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7, Problem 7.113QP
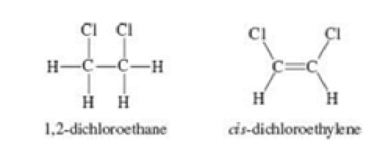
The compound 1,2-dichloroethane (C2H4Cl2) is nonpolar, while cis-dichloroethylene (C2H2Cl2) has a dipole moment. The reason for the difference is that groups connected by a single bond can rotate with respect to each other, but no rotation occurs when a double bond connects the groups. On the basis of bonding considerations, explain why rotation occurs in 1,2-dichloroethane but not in cis-dichloroethylene.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The concept of valence-shell electron-pair repulsion
(VSEPR) is presented in introductory organic chemistry
as a way to predict molecular geometries. The idea be-
hind VSEPR is that areas of electron density repel each
other so that the geometry of bonds and/or lone pairs
of electrons around any one atom places these areas as
far apart as possible. For four areas of electron density,
a tetrahedral geometry is predicted. For three areas of
electron density, a trigonal planar geometry is predicted.
Two areas of electron density lead to a linear geometry.
VSEPR is simply a predictive tool, but in some
cases, it gives an incorrect prediction. In these
instances, additional insights into bonding are
necessary, such as resonance. Interestingly, several
important situations are critical to biochemistry where
VSEPR breaks down. Two examples are shown here.
:O:
CH3
H,N
H3N
CH3 H
:0:
:0:
Histidine
The circled nitrogen atoms are predicted by VSEPR
to be tetrahedral in geometry because each…
Methylene chloride (CH2CL2) is polar, whereas carbon tetrachloride (CCl4) is nonpolar. Explain
Draw a line-bond structure for propane, CH3CH2CH3. Predict the value of each bond angle, and indicate the overall shape of the molecule.
Chapter 7 Solutions
Chemistry: Atoms First (Comp. Instructor's)
Ch. 7.1 - Determine the shapes of (a) SO3 and (b) ICl4.Ch. 7.1 - Determine the shapes of (a) CO2 and (b) SCl2.Ch. 7.1 - (a) From what group must the terminal atoms come...Ch. 7.1 - These four models may represent molecules or...Ch. 7.1 - Acetic acid, the substance that gives vinegar its...Ch. 7.1 - Ethanolamine (HOCH2CH2NH2) has a smell similar to...Ch. 7.1 - The bond angle in NH3 is significantly smaller...Ch. 7.1 - Which of these models represents a species in...Ch. 7.1 - What are the electron-domain geometry and...Ch. 7.1 - What are the electron-domain geometry and...
Ch. 7.1 - Prob. 7.1.3SRCh. 7.1 - Prob. 7.1.4SRCh. 7.2 - Prob. 7.3WECh. 7.2 - Prob. 3PPACh. 7.2 - For each of the following hypothetical molecules,...Ch. 7.2 - Which of these models could represent a polar...Ch. 7.2 - Prob. 7.2.1SRCh. 7.2 - Prob. 7.2.2SRCh. 7.3 - Prob. 7.4WECh. 7.3 - Prob. 4PPACh. 7.3 - Prob. 4PPBCh. 7.3 - Prob. 4PPCCh. 7.3 - Prob. 7.3.1SRCh. 7.3 - Which of the following exhibits significant...Ch. 7.4 - Hydrogen selenide (H2Se) is a foul-smelling gas...Ch. 7.4 - Prob. 5PPACh. 7.4 - For which molecule(s) can we not use valence bond...Ch. 7.4 - Which of these models could represent a species...Ch. 7.4 - Prob. 7.4.1SRCh. 7.4 - Prob. 7.4.2SRCh. 7.5 - Prob. 7.6WECh. 7.5 - Use hybrid orbital theory to describe the bonding...Ch. 7.5 - Prob. 6PPBCh. 7.5 - Prob. 6PPCCh. 7.5 - Prob. 7.5.1SRCh. 7.5 - Prob. 7.5.2SRCh. 7.6 - Thalidomide (C13H10N2O4) is a sedative and...Ch. 7.6 - The active ingredient in Tylenol and a host of...Ch. 7.6 - Determine the total number of sigma and pi bonds...Ch. 7.6 - In terms of valence bond theory and hybrid...Ch. 7.6 - In addition to its rise in aqueous solution as a...Ch. 7.6 - Use valence bond theory and hybrid orbitals to...Ch. 7.6 - Use valence bond theory and hybrid orbitals to...Ch. 7.6 - Explain why hybrid orbitals are necessary to...Ch. 7.6 - Prob. 7.6.1SRCh. 7.6 - Prob. 7.6.2SRCh. 7.6 - Prob. 7.6.3SRCh. 7.6 - Prob. 7.6.4SRCh. 7.7 - Prob. 7.9WECh. 7.7 - Use molecular orbital theory to determine whether...Ch. 7.7 - Use molecular orbital theory to determine whether...Ch. 7.7 - For most of the homonuclear diatomic species shown...Ch. 7.7 - Calculate the bond order of N22+, and determine...Ch. 7.7 - Which of the following species is paramagnetic?...Ch. 7.7 - Prob. 7.7.3SRCh. 7.7 - Prob. 7.7.4SRCh. 7.8 - It takes three resonance structures to represent...Ch. 7.8 - Use a combination of valence bond theory and...Ch. 7.8 - Use a combination of valence bond theory and...Ch. 7.8 - Which of the following contain one or more...Ch. 7.8 - Which of the atoms in BCl3 need hybrid orbitals to...Ch. 7.8 - Which of the following can hybrid orbitals be used...Ch. 7.8 - Which of the following enables us to explain the...Ch. 7 - Prob. 7.1KSPCh. 7 - Which of the following species does not have...Ch. 7 - Prob. 7.3KSPCh. 7 - Prob. 7.4KSPCh. 7 - Prob. 7.1QPCh. 7 - Sketch the shape of a linear triatomic molecule, a...Ch. 7 - Prob. 7.3QPCh. 7 - Prob. 7.4QPCh. 7 - In the trigonal bipyramidal arrangement, why does...Ch. 7 - Prob. 7.6QPCh. 7 - Predict the geometry of the following molecules...Ch. 7 - Prob. 7.8QPCh. 7 - Predict the geometries of the following species...Ch. 7 - Predict the geometries of the following ions: (a)...Ch. 7 - Prob. 7.11QPCh. 7 - Prob. 7.12QPCh. 7 - Prob. 7.13QPCh. 7 - Describe the geometry about each of the central...Ch. 7 - Prob. 7.15QPCh. 7 - Prob. 7.16QPCh. 7 - Prob. 7.17QPCh. 7 - Prob. 7.18QPCh. 7 - Prob. 7.19QPCh. 7 - Prob. 7.20QPCh. 7 - Prob. 7.21QPCh. 7 - Prob. 7.22QPCh. 7 - Explain the term polarizability. What kind of...Ch. 7 - Prob. 7.24QPCh. 7 - What physical properties are determined by the...Ch. 7 - Prob. 7.26QPCh. 7 - Describe the types of intermolecular forces that...Ch. 7 - The compounds Br2 and ICl are isoelectronic (have...Ch. 7 - If you lived in Alaska, which of the following...Ch. 7 - The binary hydrogen compounds of the Group 4A...Ch. 7 - List the types of intermolecular forces that exist...Ch. 7 - Prob. 7.32QPCh. 7 - Prob. 7.33QPCh. 7 - Prob. 7.34QPCh. 7 - Diethyl ether has a boiling point of 34.5C, and...Ch. 7 - Prob. 7.36QPCh. 7 - Which substance in each of the following pairs...Ch. 7 - Prob. 7.38QPCh. 7 - What kind of attractive forces must be overcome to...Ch. 7 - Prob. 7.40QPCh. 7 - Prob. 7.41QPCh. 7 - The following compounds have the same molecular...Ch. 7 - Prob. 7.43QPCh. 7 - Prob. 7.44QPCh. 7 - Use valence bond theory to explain the bonding in...Ch. 7 - Prob. 7.46QPCh. 7 - Prob. 7.47QPCh. 7 - Prob. 7.48QPCh. 7 - Prob. 7.49QPCh. 7 - What is the hybridization of atomic orbitals? Why...Ch. 7 - Prob. 7.51QPCh. 7 - Prob. 7.52QPCh. 7 - Prob. 7.53QPCh. 7 - Describe the bonding scheme of the AsH3 molecule...Ch. 7 - Prob. 7.55QPCh. 7 - Prob. 7.56QPCh. 7 - Describe the hybridization of phosphorus in PF5.Ch. 7 - Prob. 7.58QPCh. 7 - Prob. 7.59QPCh. 7 - Prob. 7.1VCCh. 7 - Prob. 7.2VCCh. 7 - Prob. 7.3VCCh. 7 - Prob. 7.4VCCh. 7 - Prob. 7.60QPCh. 7 - Which of the following pairs of atomic orbitals of...Ch. 7 - Prob. 7.62QPCh. 7 - Prob. 7.63QPCh. 7 - Prob. 7.64QPCh. 7 - Prob. 7.65QPCh. 7 - Prob. 7.66QPCh. 7 - Prob. 7.67QPCh. 7 - Prob. 7.68QPCh. 7 - Benzo[a]pyrene is a potent carcinogen found in...Ch. 7 - What is molecular orbital theory? How does it...Ch. 7 - Define the following terms: bonding molecular...Ch. 7 - Prob. 7.73QPCh. 7 - Prob. 7.74QPCh. 7 - Prob. 7.75QPCh. 7 - Draw a molecular orbital energy level diagram for...Ch. 7 - Prob. 7.77QPCh. 7 - Prob. 7.78QPCh. 7 - Prob. 7.79QPCh. 7 - Acetylene (C2H2) has a tendency to lose two...Ch. 7 - Compare the Lewis and molecular orbital treatments...Ch. 7 - Prob. 7.82QPCh. 7 - Prob. 7.83QPCh. 7 - Prob. 7.84QPCh. 7 - Prob. 7.85QPCh. 7 - Draw the molecular orbital diagram for the cyanide...Ch. 7 - Given that BeO is diamagnetic, use a molecular...Ch. 7 - Prob. 7.88QPCh. 7 - Prob. 7.89QPCh. 7 - Both ethylene (C2H4) and benzene (C6H6) contain...Ch. 7 - Chemists often represent benzene with the...Ch. 7 - Determine which of these molecules has a more...Ch. 7 - Nitryl fluoride (FNO2) is used in rocket...Ch. 7 - Describe the bonding in the nitrate ion NO3 in...Ch. 7 - Prob. 7.95QPCh. 7 - Prob. 7.96QPCh. 7 - Prob. 7.97QPCh. 7 - Prob. 7.98QPCh. 7 - Prob. 7.99QPCh. 7 - Antimony pentafluoride (SbF5) combines with XeF4...Ch. 7 - Prob. 7.101QPCh. 7 - The molecular model of nicotine (a stimulant) is...Ch. 7 - Predict the bond angles for the following...Ch. 7 - The germanium pentafluoride anion (GeF5) has been...Ch. 7 - Draw Lewis structures and give the other...Ch. 7 - Which figure best illustrates the hybridization of...Ch. 7 - Prob. 7.107QPCh. 7 - Prob. 7.108QPCh. 7 - Prob. 7.109QPCh. 7 - Prob. 7.110QPCh. 7 - Prob. 7.111QPCh. 7 - Cyclopropane (C3H6) has the shape of a triangle in...Ch. 7 - The compound 1,2-dichloroethane (C2H4Cl2) is...Ch. 7 - Prob. 7.114QPCh. 7 - Prob. 7.115QPCh. 7 - Prob. 7.116QPCh. 7 - Prob. 7.117QPCh. 7 - Prob. 7.118QPCh. 7 - The amino acid selenocysteine is one of the...Ch. 7 - Prob. 7.120QPCh. 7 - Prob. 7.121QPCh. 7 - Prob. 7.122QPCh. 7 - Gaseous or highly volatile liquid anesthetics are...Ch. 7 - Prob. 7.124QPCh. 7 - Prob. 7.125QPCh. 7 - Two of the drugs that are prescribed for the...Ch. 7 - Prob. 7.127QPCh. 7 - Prob. 7.128QPCh. 7 - The BO+ ion is paramagnetic. Determine (a) whether...Ch. 7 - Use molecular orbital theory to explain the...Ch. 7 - Which best illustrates the change in geometry...Ch. 7 - Prob. 7.132QPCh. 7 - Prob. 7.133QPCh. 7 - Aluminum trichloride (AlCl3) is an...Ch. 7 - Prob. 7.135QPCh. 7 - Prob. 7.136QPCh. 7 - Prob. 7.137QPCh. 7 - Consider an N2 molecule in its first excited...Ch. 7 - The Lewis structure for O2 is Use molecular...Ch. 7 - Draw the Lewis structure of ketene (C2H2O) and...Ch. 7 - The compound TCDD, or...Ch. 7 - Name the kinds of attractive forces that must be...Ch. 7 - Carbon monoxide (CO) is a poisonous compound due...Ch. 7 - Prob. 7.144QPCh. 7 - Prob. 7.145QP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Gamma hydroxybutyric acid, GHB, infamous as a date rape drug, is used illicitly because of its effects on the nervous system. The condensed molecular formula for GHB is HO(CH2)3COOH. (a) Write the Lewis structure for GHB. (b) Identify the hybridization of the carbon atom in the CH2 groups and of the terminal carbon. (c) Is hydrogen bonding possible in GHB? If so, write Lewis structures to illustrate the hydrogen bonding. (d) Which carbon atoms are involved in sigma bonds? In pi bonds? (e) Which oxygen atom is involved in sigma bonds? In pi bonds?arrow_forwardAspirin, or acetylsalicylic acid, has the formula C9H8O4 and the skeleton structure (a) Complete the Lewis structure and give the number of bonds and bonds in aspirin. (b) What is the hybridization about the CO2H carbon atom (colored blue)? (c) What is the hybridization about the carbon atom in the benzene-like ring that is bonded to an oxygen atom (colored red)? Also, what is the hybridization of the oxygen atom bonded to this carbon atom?arrow_forwardThe sulfamate ion, H2NSO3, can be thought of as having been formed from the amide ion, NH2, and sulphur trioxide, SO3. (a) What are the electron-pair and molecular geometries or the amide ion and or SO3? What are the hybridizations of the N and S atoms, respectively? (b) Sketch a structure for the sulfamate ion, and estimate the bond angles. (c) What changes in hybridization do you expect for N and S in the course of the reaction NH2 + SO3 H2NSO3? (d) Is SO3 the donor of an electron pair or the acceptor of an electron pair in the reaction with amide ion? Does the electrostatic potential map shown below confirm your prediction?arrow_forward
- Use the VSEPR model to predict the bond angles around each central atom in the following Lewis structures (benzene rings are frequently pictured as hexagons, without the letter for the carbon atom at each vertex). Note that the drawings do not necessarily depict the bond angles correctly.arrow_forwardBenzene is an organic chemical compound with the molecular formula C6Hỏ. In the benzene molecule, carbon atoms form a ring with alternating single and double bonds connecting them. Thus, each individual carbon atom forms one o bond with another carbon atom and one σ and one π bond with another carbon atom. Each carbon atom also forms one o bond with a hydrogen atom. H H Identify which types of orbitals overlap to form the bonds between the atoms in a benzene molecule. Drag the appropriate labels to their respective targets. View Available Hint(s) р-р sp² - sp² sp³ – sp³ sp-sp sp² - s Bonds formed Carbon-carbon o bond Carbon-carbon bond H-C Carbon-hydrogen o bond H C-C 000 C=C Type of orbitals involved C-H H Reset Helparrow_forwardOctahedral molecules only have a single bond angle - write down its value. What angle do both the square pyramid and square planar structures contain?The trigonal planar (1) and trigonal pyramid (4) structures both contain 4 (1 central and 3 apical) atoms. From the models you have created and drawn, are these two structures the same? If they are not, identify the key angles which distinguish these two structures.arrow_forward
- Allene, CH2 “C “CH2, has the structure shown below. Explain how the bonding in allene requires the two “CH2 groups at its ends to be at right angles to each other.arrow_forward3 2 4 5 Consider the following second-period heteronuclear diatomic molecule. Combining the valence orbitals (2px, 2py, 2pz, and 2s) of each of the two atoms creates eight new orbitals (one 02s*, one 02s, one 02p, one 02p, two л₂p, and two л₂p) that span the entire molecule and explain it's electronic properties. Those eight new orbitals exist at six energy levels. Rank the molecular orbitals of a F2 molecule from lowest (1) to highest (6) energy. Fatom 6- highest 1- lowest F2 molecule [Choose ] [Choose ] [Choose ] [Choose ] [Choose ] [Choose ] F atomarrow_forwardPart A Benzene is an organic chemical compound with the molecular formula C6H6. In the benzene molecule, carbon atoms form a ring with alternating single and double bonds connecting them. Thus, each individual carbon atom forms one o bond with another carbon atom and one o and one bond with another carbon atom. Each carbon atom also forms one o bond with a hydrogen atom. H H sp³-sp³ H Identify which types of orbitals overlap to form the bonds between the atoms in a benzene molecule. Drag the appropriate labels to their respective targets. ► View Available Hint(s) sp-sp sp² - sp² р-р Bonds formed Carbon-carbon o bond Carbon-carbon 7 bond H-C Carbon-hydrogen o bond C=C C-H H Type of orbitals involved P Pearson Reset Helparrow_forward
- Many important compounds in the chemical industry are derivatives of ethylene(C2H4). two of them are acrylonitrile and methyl methacrylate.AcrylonitrileMethyl methacrylateComplete the Lewis structures, showing all lone pairs. Give approximate values forbond angles a through f Give the hybridization of all carbon atoms. In acrylonitrile,how many of the atoms in the molecule must lie in the same plane? How many σbonds and how many π bonds are there in methyl methacrylate and acrylonitrile?arrow_forwardHere's a wild molecule: disulfur dinitride (S2N2). It can be isolated as a crystalline solid, but it is unstable (explosive). Therefore, it must to be handled carefully in an inert atmosphere free of oxygen. X-ray diffraction measurements of the crystalline solid indicate the atoms are arranged in a square with the sulfur atoms on opposite corners and the nitrogen atoms on the remaining opposite corners. Consider the bonding in this unusual molecule. a. Determine the best Lewis structure representation for S2N2. Include the formal charges on each atom in the structure. b. Determine the valence bonding theory predictions for hybridization at each atom in S2N2. The bond angles in S2N2 have been measured by X-ray diffraction and are all approximately 90°. Does valence bond theory do a good job of predicting this observation? Consider the geometry and bonding of S2N2 computed using molecular orbital theory. The results are shown below. Identify the orbital character of the HOMO(-2),…arrow_forwardVISUALIZATION Energy Diagram for Conversion of Cis-2-Butene to Trans-2-Butene 600 500 400 Reactant 300 200 E = 435 x 1021 J 100 Initial state AE-7 X 10 J -30° 30 60 90° 120° 150° 180° 210° Reaction progress (angle of twist) Cis-2-butene is one of two forms of butene where the middle two carbon atoms are joined by a double bond. The other form is trans-2-butene. The conversion of the cis form to the trans form is slow at room temperature, and the two forms represent two distinct compounds. Conversion of the cis to the trans form involves rotation about the C-C double bond, which requires a substantial amount of energy on the molecular scale. Move the slider to observe the different conformations and their energies along the pathway of the conversion. Potential Energy 1021 Jarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY