
Concept explainers
The chapter sections to review are shown in parentheses at the end of each problem.
Using the models of the molecules (black = C, white = H, yellow = S, green = Cl), determine each of the following for models of compounds 1 and 2: (7.2, 7.4, 7.5, 7.6)
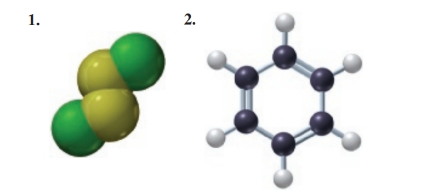
a. molecular formula
c. molar mass
b. empirical formula
d. mass percent composition
(a)
Interpretation: To find the empirical formula of dipyrithione.
Concept Introduction: Empirical formula is the simplest ratio of different elements in a compound.
Answer to Problem 61UTC
The empirical formula of dipyrithione is C5H4NOS
Explanation of Solution
The molecular formula of dipyrithione is C10H8N2O2S2 The subscript of each element is divided by 2. 2 is the minimum number of atoms of elements N, O and S.
Thus the empirical formula is C5H4NOS
(b)
Interpretation: To find the molar mass of dipyrithione.
Concept Introduction: Molar mass is found by adding the atomic mass of each element that makes up the compound.
Answer to Problem 61UTC
Molar mass of dipyrithione = 252 g/mol
Explanation of Solution
The molecular formula of dipyrithione is C10H8N2O2S2 Atomic mass of C = 12 g/mol
Atomic mass of H = 1 g/mol
Atomic mass of N = 14 g/mol
Atomic mass of O = 16 g/mol
Atomic mass of S = 32 g/mol
Molar mass of dipyrithione, C10H8N2O2S2 =
(c)
Interpretation: To find the mass percent of O in dipyrithione.
Concept Introduction:
Answer to Problem 61UTC
Mass percent of O in dipyrithione = 12.7 %
Explanation of Solution
The molecular formula of dipyrithione is C10H8N2O2S2 Since from the molecular formula of dipyrithione it’s clear that dipyrithione contains 2 atoms of oxygen so, Mass percent of O in dipyrithione =
(d)
Interpretation: To find the grams of C in 2.50 g of dipyrithione.
Concept Introduction: An arithmetical multiplier which is used for converting a quantity expressed in one unit into another equivalent set of units is said to be conversion factor.
Answer to Problem 61UTC
11.9 g of C is present in 2.50 g of dipyrithione.
Explanation of Solution
The molecular formula of dipyrithione is C10H8N2O2S2 Since from the molecular formula of dipyrithione it’s clear that dipyrithione contains 10 atoms of carbon so,
Thus,
Hence, the mass of carbon in 25.0 g of dipyrithione is 11.9 g.
(e)
Interpretation: To find the moles of dipyrithione in 25.0 g of dipyrithione.
Concept Introduction:
The relationship between mass, moles, and molar mass:
Answer to Problem 61UTC
Moles of dipyrithione = 0.0992 mol
Explanation of Solution
The relationship between mass, moles, and molar mass:
Mass of dipyrithione is 25.0 g (given)
Molar mass of dipyrithione is 252 g/mol.
Moles of dipyrithione =
Want to see more full solutions like this?
Chapter 7 Solutions
Pearson eText Basic Chemistry -- Instant Access (Pearson+)
- (6.3)The volume of a gas is 0.953 L at 30.0 °C. If the gas is heated to 60.0 °C, what would be the volume of the gas (in L) at this temperature? O 1.05 L O 0.867 L O 1.91 L O 0.477 Larrow_forwardOne of the reactions in the industrial production ofnitric acid involves the production of nitric oxide: 4 NH3(g) 1 5 O2(g) → 4 NO(g) 1 6 H2O(g) (7.4) T/I a) If 4500 kg of ammonia, NH3(g), react with7500 kg of O2, what mass of NO will form? (b) What mass of the excess reagent will remain?arrow_forward1. (10.3- 1.24)/18.43 2. A penny minted after 1982 has a mass of 2.5 g Calculate total mass of three such pennies. 3. The following are placed in a beaker weighing 39.457 g: 2.689 g of NaCl, 1.26 g of sand and 5.0 g water. What is the final mass of the beaker? 4. If the beaker containing a sample of alcohol weighs 49.8767 g and the empty beaker weighs 49.214 g, what is the weight of the alcohol? 5. A flask that weighs 345.8 g is filled with 225 mL of carbon tetrachloride. The weight of the flask and carbon tetrachloride is found to be 703.55 g. From this information, calculate the density of carbon tetrachloride.arrow_forward
- Honors Chemistry-4th Hour- Dr. Paul / Gases / Lesson 148 11. Which state of matter is most accurately described by the kinetic molecular theory? O gas aqueous solid O liquid available! (3.0.227)arrow_forward(3.6)What is the formula of the compound that forms between Pb4+ ion and sulfite ion? Pb₂SO3 O Pb2(SO3)4 O Pb(SO3)2 O Pb4(SO3)2 << Previousarrow_forward6.149 Indicate the major type of intermolecular forces-(1) ionic bonds, (2) dipole-dipole attractions, (3) hydrogen bonds, (4) dispersion forces-that occurs between particles of the following: (6.9) a. NF3 d. Cs,0 с. Brz f. CH;OH b. CIF e. C4H10arrow_forward
- hemistry-4th Hour - Dr. Paul / Gases/Lesson 148 Honor 22. According to Charles' law, what kind of relationship does temperature and volume have? O direct relationship O no relationship O indirect relationship chemical relationship GET IT ablo! (3.0.227)arrow_forward7. What type of product is formed when acids are added to some ionic compounds? Stronger acids Weaker acids Gas Solids GET New version available! (3.0.220)arrow_forward11. What type of product is formed when acids are added to some ionic compounds? Stronger acids Solids Gas Weaker acids New version available! (3.0.220) GET IT NOWarrow_forward
- (3.9)Calculate the percent composition of sulfur in aluminum sulfate, Al2(SO4)3. 28.1% 17.7% 71.9% 33.6% ◄ Previousarrow_forward9.2 An unknown salt is found to contain 30,6% of water. The formula mass for anhydrous salt (AS) is 100 amu. 9.2.1 Calculate the amount of water bonded with an unknown salt. 9.2.2 An anhydrous salt above consists of a metal with a molar mass of 40. (a) Write down the chemical symbol of this metal. (b) To which group does the metal belongs? 9.2.3 Write down the chemical formula of an anhydrous salt above.arrow_forwardA student sets up and solves the following equation to solve a problem in solution stoichiometry. Fill in the missing part of the student's equation. (47.6) 65.08 mol 1 mL -3 10 L mol (3²) 3.2 L = 2.3 x 10 mL x10 ロ・ロ X μarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





