
An important parameter in the design of gas absorbers is the ratio of the ?ow rate of the feed liquid to that of the feed gas. The lower the value of this ratio, the lower the cost of the solvent required to process a given quantity of gas but the taller the absorber must be to achieve a speci?ed separation.
Propane is recovered from a 7 mole% propane—93% nitrogen mixture by contacting the mixture with liquid n-decane. An insigni?cant amount of decane is vaporized in the process, and 98.5% of the propane entering the unit is absorbed.
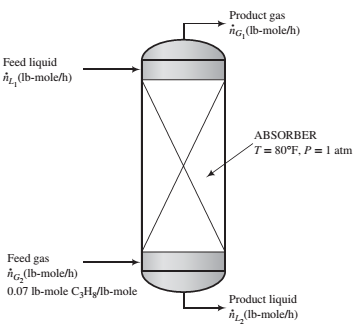
(a) The highest possible propane mole fraction in the exiting liquid is that in equilibrium with the propane mole fraction in the feed gas (a condition requiring an in?nitely tall column). Using Raoult’s law to relate the mole fractions of propane in the feed gas and liquid, calculate the ratio 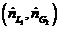 corresponding to this limiting condition.
corresponding to this limiting condition.
(b) Suppose the actual feed ratio 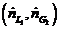 is 1.2 times the value calculated in Part (a) and the percentage of the entering propane absorbed is the same (98.5%). Calculate the mole fraction of propane in the exiting liquid.
is 1.2 times the value calculated in Part (a) and the percentage of the entering propane absorbed is the same (98.5%). Calculate the mole fraction of propane in the exiting liquid.
(c) What are the costs and bene?ts associated with increasing 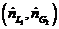 from its minimum value [the value calculated in Part (a)]? What would you have to know to determine the most cost−effective value of this ratio?
from its minimum value [the value calculated in Part (a)]? What would you have to know to determine the most cost−effective value of this ratio?
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
ELEMENTARY PRINCIPLES OF CHEM. PROCESS.
Additional Science Textbook Solutions
Elements of Chemical Reaction Engineering (5th Edition) (Prentice Hall International Series in the Physical and Chemical Engineering Sciences)
Process Dynamics and Control, 4e
Computer Science: An Overview (12th Edition)
Starting Out with Python (3rd Edition)
Automotive Technology: Principles, Diagnosis, And Service (6th Edition) (halderman Automotive Series)
Digital Fundamentals (11th Edition)
- Leaching of Oil from Soybeans in a Single Stage. Repeat Example 12.9-1 for single-stage leaching of oil from soybeans. The 100 kg of soybeans contains 22 wt % oil and the solvent feed is 80 kg of solvent containing 3 wt % soybean oi!. = 0.239, V, = 50.0 kg, x41 = 0.239, N, = 1.5 Ans. L, = 52.0 kg, ya1 wwwwwarrow_forwardO 15 Ppm 2. In a certain polluted area, the partial pressure of NO gas was 8 pascal. The mixing ratio of NO is . ppmv (assume normal conditions) 3. A 500 kg sample of a fertilizer containing 115 ppms Cu was mixed with 15,000 kg soil which also contains 2.0 ppms Cu. The final level of Cu is ........ pPpms 4. The mixing ratio of H,S gas is 0.2 ppmv and the total flux into atmosphere is 2.5x10" kg Estimate r for H;S. ....kg (assume normal conditions and volume of atmosphere 4.0x10" m)arrow_forward18. Jet is an undergraduate chemistry student, he’s out in the laboratory trying to determine the volatile organic compounds as well as overall protein content of the leaf and stem of a malunggay (Moringa oleifera). He subjected the leaf and stem in a separate digestion reaction (treatment of sulfuric acid), afterwards he subjected the products to high temperature induction to get a dry ash like substance. Which type/s of chemical analysis did Jet employ to reach his objective?I. Qualitative AnalysisII. Quantitative Analysis III. Instrumental Chemical AnalysisIV. Wet Chemical AnalysisA. II & IV onlyB. I & IV onlyC. I, II & IV onlyD. I, II, III, IVarrow_forward
- 1) 1500 kg mixture contains 15% acetone, 75% water and 10% Acetic acid by weight. a) Calculate the total mole of mixture and molar fractions of each compound. b) Calculate Average Molecular Weight of mixture. c) 2 kmol more Acetic acid is added to the mixture. What would be the mass fractions of this new mixture? (MW: Acetone= 58 g/mol Water= 18 g/mol Acetic acid= 60 g/mol)arrow_forwardb) A gaseous mixture contain 32.9 mol % He, 40.7 mol % N2 and 26.4 mol % Ar. Determine the composition of this mixture on a mass basis based on the molecular weight given: MW (He) = 4.003 , MW (N2) = 28.02, MW(Ar) = 39.95 %3Darrow_forwardThank youarrow_forward
- A sample of enzyme involved in alcohol metabolism, extracted and purified extensively from yeast cultures, was loaded onto a gel filtration column for molecular weight approximation. A set of protein standards with known molecular weight was eluted separately through the same column and suitable buffer following the elution of blue dextran for void volume determination. The following table summarizes the data obtained from the experiment: Elution flow rate 1 mL/min Sample collector setting Continuous; Sequential Container change rate 1 tube/min Sample/Standard eluted Test tube No/s. MW (Da) Blue dextran 4 2,000,000 Bovine thyroglobulin 11 669,000 Horse spleen apoferritin 14 443,000 Beta-amylase from sweet potato 21 200,000 Bovine serum albumin 27 66,000 Alcohol metabolism enzyme 19 ? 1. Process the given raw data in a tabular format of properly identified data points ready for calibration curve plotting. 2. Construct the calibration curve using MS Excel for use in MW determination.…arrow_forward3. Determine the porosity of a sweet Vidali onion, which is composed of 93.8% water, 1.04% protein, 0.45% fat, 4.48% carbohydrate, and 0.46% ash. The apparent density of the onion was measured by the liquid displacement method. Toulene was used as the pycnometer liquid. All the measurements were carried out at 20°C and the density of the toluene at that temperature is known to be 865 kg/m³. The date obtained from liquid displacement measurements are: Weight of the empty pycnometer: Weight of the pycnometer filled with toluene: Weight of the pycnometer containing only onion: Weight of the pycnometer containing the onion and toluene: 75.87 g 126.58 g 85.87 g 127.38 garrow_forwardCalculate the solubility at 25 °C of PbCO3 in pure water and in a 0.0100M Pb(NO3), solution. You'll find Ksp data in the ALEKS Data tab. Round both of your answers to 2 significant digits. 0 // solubility in pure water: x10 solubility in 0.0100 M Pb(NO3)2 0-/- solution: L X 09 00 X S ?arrow_forward
- For the purpose of determination of agricultural pesticides in river water, 3.2 liters of river water were taken and extracted with 125ml of CCI4 solvent using a separating funnel. After shaking and reaching the state of equilibrium, if you know that the value of the distribution coefficient (KD) is equal to 20, the weight ratio of the pesticides in the boiling layer to the lower layer in the separating funnel?arrow_forward6. Assume you have 1kg of an alloy consisting of 85 wt% Sn and 15 wt% Pb at a temperature of 184 °C. How much Sn (in kg) needs to be added to solidify the entire mixture without lowering the temperature? Composition (at% Sn) 20 40 60 80 100 327°C Temperature (°C) 300 200 100 (Pb) a + L 18.3 20 183°C a + B Liquid 61.9 40 60 Composition (wt% Sn) B+L 80 232°C 97.8 B al 600 500 400 300 200 100 100 (Sn) Temperature (°F)arrow_forwardChemistry The levels of an organic pollutant (P) in the groundwater at the perimeter of a plant were a cause for concern. A 10 mL sample of the water was taken and the pollutant was extracted with 95% efficiency using 25 mL of diethyl ether. GC was used to analyse the concentration of P in diethyl ether. A calibration curve was plotted for a series of standards of P which yielded the following results: Peak Area Toluene Conc. (µg/ml) 12,000 2.6 23,700 5.0 35,500 7.7 46,800 9.9 31,250 Sample Determine the concentration of P in ppb in the initial groundwater sample.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





