
Concept explainers
(a)
Interpretation:
The given compound should be prepared using
Concept Introduction:
Addition of
Catalytic hydrogenation in presence of
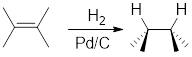
For cyclic reactants, the addition of
Enantiomers: they are chiral molecules whose mirror images are not superimposable.
Enantiomers are given configuration as R or S based on the atoms bonded with chiral (carbon bonded with four different substituents) carbon.
Carbocation: carbon ion that bears a positive charge on it.
Carbocation stability order:

Nucleophile: It donates pair of electrons to positively charged substrate resulting in the formation of
Electrophile: It is positively charged species which seeks for negative charge and hence accepts pair of electrons from negatively charged species (Nucleophiles) which results in the formation of chemical bond.
(b)
Interpretation:
The given compound should be prepared using alkene and other reagents.
Concept Introduction:
Addition of
Catalytic hydrogenation in presence of
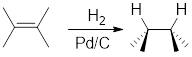
For cyclic reactants, the addition of
Enantiomers: they are chiral molecules whose mirror images are not superimposable.
Enantiomers are given configuration as R or S based on the atoms bonded with chiral (carbon bonded with four different substituents) carbon.
Carbocation: carbon ion that bears a positive charge on it.
Carbocation stability order:

Nucleophile: It donates pair of electrons to positively charged substrate resulting in the formation of chemical bond.
Electrophile: It is positively charged species which seeks for negative charge and hence accepts pair of electrons from negatively charged species (Nucleophiles) which results in the formation of chemical bond.
(c)
Interpretation:
The given compound should be prepared using alkene and other reagents.
Concept Introduction:
Addition of hydrogen halides:
In the addition of hydrogen halides over alkenes, first the
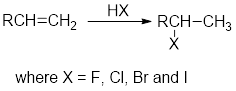
Carbocation: carbon ion that bears a positive charge on it.
Carbocation stability order:

Nucleophile: It donates pair of electrons to positively charged substrate resulting in the formation of chemical bond.
Electrophile: It is positively charged species which seeks for negative charge and hence accepts pair of electrons from negatively charged species (Nucleophiles) which results in the formation of chemical bond.
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
EBK ESSENTIAL ORGANIC CHEMISTRY
- Using your reaction roadmap as a guide, show how to convert cyclohexane into hexanedial. Show all reagents and all molecules synthesized along the way.arrow_forward2) How would you synthesize the following compounds from cyclohexanone? CH₂Br A Br B CH₂C6H5 D CH2CH2CO2Harrow_forwardEthylene oxide is the starting material for the synthesis of 1,4-dioxane. Propose a mechanism for each step in this synthesis.arrow_forward
- Synthesize the following compound from cyclohexanone and any other organic and inorganic reagents you need.arrow_forward2) How would you synthesize the following compounds from cyclohexanone? CH₂Br CH₂C6H5 A Br B C D CH,CH2CO2Harrow_forwardSynthesize the following compound from cyclohexanone and any other organic and inorganic reagents you need.arrow_forward
- How does propane synthesized from propanone? Show its mechanism.arrow_forwardHow would you synthesize the followingcompounds from benzene using thereagents from the table? Each requires 3-4steps.arrow_forwardb) The Wolf-Kishner reduction is a reaction used in Organic Chemistry to convert carbonyl functionalities into methylene group. The reaction was used to convert an aldehyde or ketone to an alkane using hydrazine, base and thermal conditions. The mechanism begins with the attack of hydrazine of the aldehyde or ketone. Stage 1: The reaction of aldehyde/ketone with hydrazine to produce hydrazine Stage 2: Reaction with the base and heat to convert hydrozone to alkane Write the mechanism of the reaction.arrow_forward
- How can you synthesize the following compound using 2-methylbutane as the starting material?arrow_forwardComplete the following multistep syntheses by showing the major products and the reagents nessesary for each steparrow_forward5. Synthesize the following compounds by using cyclohexanone and propene as your only sources of carbon. Any other needed reagents are available.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

