
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5.10C, Problem 5.19P
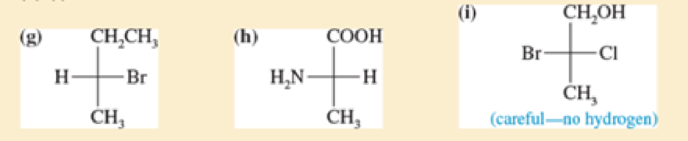
For each Fischer projection, label each asymmetric carbon atom as (R) or (S) (a)-(f) the structures in Problem5-18
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Locate with an asterisk the stereogenic centers (if any) in the following structure:
B. C6H5CH2CH(OH)CO2H
The structure of 4 isomers of ketopentose are shown.
1) select every structure that is a diastereomer of structure D
A, B, or C?
2) select every structure that is a enantiomer of structure C
A, B, or D
3) select every structure that is a stereoisomer of structure C
A, B, or D
Draw each compound in its most symmetric conformation, star (*) any asymmetric carbon atoms, and draw any mirror planes.Label any meso compounds. You may use Fischer projections if you prefer.(a)(R, R)-2,3-dibromobutane
Chapter 5 Solutions
Organic Chemistry (9th Edition)
Ch. 5.2 - Determine whether the following objects are chiral...Ch. 5.2A - Prob. 5.2PCh. 5.2B - Prob. 5.3PCh. 5.2B - Prob. 5.4PCh. 5.2C - Prob. 5.5PCh. 5.3 - Prob. 5.6PCh. 5.3 - Prob. 5.7PCh. 5.4D - Prob. 5.8PCh. 5.4D - Prob. 5.9PCh. 5.4D - Prob. 5.10P
Ch. 5.5 - Prob. 5.11PCh. 5.7 - When optically pure (R)-2-bromobutane is heated...Ch. 5.7 - Prob. 5.13PCh. 5.8 - Prob. 5.14PCh. 5.9B - Draw three-dimensional representations of the...Ch. 5.10A - For each sot of examples, make a model of the...Ch. 5.10A - Draw a Fischer projection for each compound....Ch. 5.10B - Prob. 5.18PCh. 5.10C - For each Fischer projection, label each asymmetric...Ch. 5.11C - Prob. 5.20PCh. 5.13 - Prob. 5.21PCh. 5.13 - Prob. 5.22PCh. 5.15 - Prob. 5.23PCh. 5.16A - Prob. 5.24PCh. 5 - The following four structures are naturally...Ch. 5 - For each structure, 1. star () any asymmetric...Ch. 5 - Prob. 5.27SPCh. 5 - Prob. 5.28SPCh. 5 - Prob. 5.29SPCh. 5 - Prob. 5.30SPCh. 5 - Prob. 5.31SPCh. 5 - Prob. 5.32SPCh. 5 - Prob. 5.33SPCh. 5 - Prob. 5.34SPCh. 5 - For each structure, 1. draw all the stereoisomers....Ch. 5 - Prob. 5.36SPCh. 5 - Prob. 5.37SPCh. 5 - 3,4-Dimethylpent-1-ene has the formula...Ch. 5 - A graduate student was studying enzymatic...Ch. 5 - Prob. 5.40SPCh. 5 - Prob. 5.41SP
Additional Science Textbook Solutions
Find more solutions based on key concepts
1. What did each of the following scientists contribute to our knowledge of the atom?
a. William Crookes
b. E...
Chemistry For Changing Times (14th Edition)
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
Determine the de Brogue wavelength of a. an electron moving at 1/10 the speed of light. b. a 400 g Frisbee movi...
Inorganic Chemistry
Draw a Lewis structure for each covalent molecule. a. HBr b. CH3F c. H2O2 d. N2H4 e. C2H6 f. CH2Cl2
Principles of General, Organic, Biological Chemistry
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Determine whether each pair of molecules represent: (a) identical compounds; (b) constitutional (structural) isomers, (c) enantiomers, or (d) diastereomers. Thank you.arrow_forwardH3C Cl \ / C=C / \ H2N I (R)-enantiomer (Z) isomer (S)-enantiomer (E) isomer meso compoundarrow_forwardRe-draw each Fischer projection formula using wedges and dashed wedgesfor the stereogenic center, and label the center as R or S.arrow_forward
- Consider CH3-CH(OH)-CH(OH)(Br). a.How many stereogeniccenters are in the molecule? b.How many stereoisomers are there for the compound? c.Draw the Fischer projection for each of the stereoisomer. Label each using I, II, etc. d.Which pairs are enantiomers? Which are diastereomers? e.Determine the absolute configuration of each chiral center in one pair of diastereomer.arrow_forwardDraw each compound in its most symmetric conformation, star (*) any asymmetric carbon atoms, and draw any mirror planes. Label any meso compounds. You may use Fischer projections if you prefer.(a) ({)-2,3-dibromo-2,3-dichlorobutanearrow_forwardA(n) ________ is an achiral compound that contains chiral centers but is superimposable on its mirror image. A) constitutional isomers B) conformational isomers C) enantiomers D) diastereomers E) meso compoundsarrow_forward
- Convert each three-dimensional representation into a Fischer projection.arrow_forwardConsider Newman projections (A–D) for four-carbon carbohydrates. Howis each pair of compounds related: (a) A and B; (b) A and C; (c) A and D; (d) C and D? Choose from identical molecules, enantiomers, or diastereomers.arrow_forwardFor each pair, indicate whether they are constitutional isomers, enantiomers, diastereoisomers, or identical molecules. A BDEFarrow_forward
- Consider Newman projections (A–D) for four-carbon carbohydrates. How is each pair of compounds related: (a) A and B; (b) A and C; (c) A and D; (d) C and D? Choose from identical molecules, enantiomers, or diastereomers.arrow_forward(a) Draw all possible stereoisomers of 4-methylnon-2-ene, and name each isomer, including its E,Z and R,S prexes. (b) Label two pairs of enantiomers. (c) Label four pairs of diastereomers.arrow_forwardConvert each Fischer projection into a three-dimensional representation with wedges and dashed bonds.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License