
Concept explainers
Successive substitution of F atoms for H atoms in the molecule NH3 produces the molecules NH2F, NHF2, and NF3.
- a. Draw Lewis structures for each of the four molecules.
- b. Using VSEPR theory, predict the geometry of each of the four molecules.
- c. Specify the polarity (polar or nonpolar) for each of the four molecules.
(a)
Interpretation:
Lewis structures for the four molecules that are formed by successive substitution of
Concept Introduction:
Lewis structure clearly depicts the bonding and nonbonding electrons in the atom. This is only partially useful for the molecule that contains one or more multiple bonds and when coordinate covalent bond is present in the molecule. For drawing Lewis structure a systematic procedure is followed. They are,
- The total number of valence electrons that is present in molecule is calculated by adding all the valence electrons of the atoms present in the molecule.
- The chemical symbols for the atoms that is present in the molecule is written in the order that they are bonded. After this a single covalent bond is placed between each atoms as two electrons.
- The nonbonding electrons are added to each atom that is bonded to the central atom so that it contains octet of electrons. For hydrogen alone the “octet” is only two electrons.
- The remaining electrons has to be placed on the central atom in the structure.
- If there is no octet of electrons present in the central atom, then use one or more pairs of nonbonding electrons that is bonded to the central atom to form double or triple bonds.
- The total number of electrons has to be counted and it has to be confirmed whether the count is same as that of the number of valence electrons that is available for bonding.
Explanation of Solution
Given molecules are
For
Given molecule is
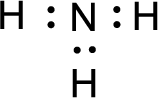
The nonbonding electrons are added to the nitrogen atom. This results in the structure as,
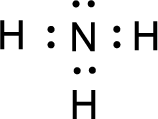
All the atoms present in the above structure contains octet of electrons. The total number of electron dots present in the above structure is 8 and it is same as the valence electrons of
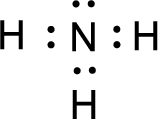
For
Given molecule is
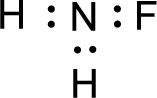
The nonbonding electrons are added to the fluorine atom and nitrogen atom. This results in the structure as,
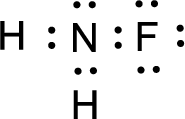
All the atoms present in the above structure contains octet of electrons. The total number of electron dots present in the above structure is 14 and it is same as the valence electrons of
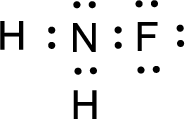
For
Given molecule is
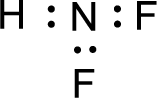
The nonbonding electrons are added to the fluorine atom and nitrogen atom. This results in the structure as,

All the atoms present in the above structure contains octet of electrons. The total number of electron dots present in the above structure is 20 and it is same as the valence electrons of

For
Given molecule is
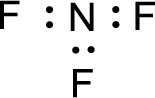
The nonbonding electrons are added to the nitrogen atom and fluorine atoms. This results in the structure as,

All the atoms present in the above structure contains octet of electrons. The total number of electron dots present in the above structure is 26 and it is same as the valence electrons of

(b)
Interpretation:
Molecule geometry has to be predicted for the four molecules using VSEPR theory.
Concept Introduction:
Information about the number of bonds and types of bonds can be obtained from Lewis structure but the molecular geometry cannot be obtained. Three dimensional arrangement of atoms in a molecule can be given by molecular geometry. Physical and chemical properties are determined by the molecular geometry of the molecule.
Using VSEPR theory and Lewis structure, the molecular geometry of the molecule that contain less number of atoms can be predicted. VSEPR theory uses the information from Lewis structure of the molecule to predict the molecular geometry of the molecule. Main concept of VSEPR theory is that electron pairs that are present in the valence shell adopt arrangement in a way that minimize the repulsion between like charges.
If the central atom contains two electron pairs, then it has to be far apart means, it has to be on opposite side of the nucleus. This means the angle has to be
If the central atom contains three electron pairs, then it has to be far apart means, it has to be on corner of a triangle. This means the angle has to be
If the central atom contains four electron pairs, then it has to be far apart means, it has to be in a tetrahedral arrangement. This means the angle has to be
The collection of valence electron that is present in localized region about central atom in a molecule is known as VSEPR electron group. This may contain two electrons, four electrons, or six electrons. The electron group that contain four and six electrons repel each other.
Tetrahedral VSEPR electron group:
The four electron pairs can be of three VSEPR electron groups. They are 4 bonding electron groups, 3 bonding and 1 nonbonding electron groups, and 2 bonding and 2 nonbonding electron groups. The molecular geometry that is associated with 4 bonding electron groups is tetrahedral. The molecular geometry that is associated with 3 bonding and 1 nonbonding electron groups is trigonal pyramidal. The molecular geometry that is associated with 2 bonding and 2 nonbonding electron groups is angular.
Trigonal planar VSEPR electron group:
The three electron pairs can be of two VSEPR electron groups. They are 3 bonding electron groups, and 2 bonding and 1 nonbonding electron groups. The molecular geometry that is associated with 3 bonding electron groups is trigonal planar. The molecular geometry that is associated with 2 bonding and 1 nonbonding electron groups is angular.
Linear VSEPR electron group:
The two electron pairs can be of only one VSEPR electron groups. It is only 2 bonding electron groups and the geometry associated with it is linear geometry.
Explanation of Solution
Given molecule is
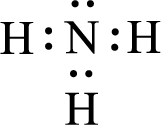
The central atom in the above molecule is found to be nitrogen. This has three bonding electron groups and one nonbonding electron groups. The arrangement around the central atom is trigonal pyramidal. Looking for molecular geometry, the central atom that contains three bonding electron groups and one nonbonding electron groups and it has trigonal pyramidal geometry as per VSEPR theory.
Given molecule is
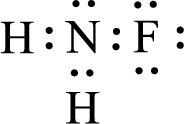
The central atom in the above molecule is found to be nitrogen. This has three bonding electron groups and one nonbonding electron groups. The arrangement around the central atom is trigonal pyramidal. Looking for molecular geometry, the central atom that contains three bonding electron groups and one nonbonding electron groups and it has trigonal pyramidal geometry as per VSEPR theory.
Given molecule is
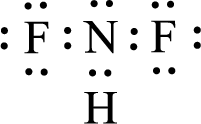
The central atom in the above molecule is found to be nitrogen. This has three bonding electron groups and one nonbonding electron groups. The arrangement around the central atom is trigonal pyramidal. Looking for molecular geometry, the central atom that contains three bonding electron groups and one nonbonding electron groups and it has trigonal pyramidal geometry as per VSEPR theory.
Given molecule is
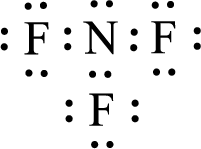
The central atom in the above molecule is found to be nitrogen. This has three bonding electron groups and one nonbonding electron groups. The arrangement around the central atom is trigonal pyramidal. Looking for molecular geometry, the central atom that contains three bonding electron groups and one nonbonding electron groups and it has trigonal pyramidal geometry as per VSEPR theory.
(c)
Interpretation:
The four molecules that are given has to be classified as polar and nonpolar.
Concept Introduction:
Measure of the degree of inequality in attraction of the bonding electrons to the various locations present within a molecule is known as molecular polarity. This can also be said in terms of electron attraction and that is in a molecule one part is favored than the other parts of the molecule.
If in a molecule there is an uneven distribution of electronic charges means it is known as polar molecule. If there is a symmetrical distribution of electron charge over the molecule means it is known as nonpolar molecule. Two factors that decide molecular polarity is bond polarity and geometry of molecule. If a molecule is symmetrical means then there won’t be any molecular polarity because the effect given by the polar bonds may cancel out each other.
The polarity of the bonds, arrangement of the bonds determines the degree of molecular polarity. If the electronegativity difference is more, then the molecule will be more polar.
Explanation of Solution
Given molecules are
Want to see more full solutions like this?
Chapter 5 Solutions
General, Organic, And Biological Chemistry, Hybrid (with Owlv2 Quick Prep For General Chemistry Printed Access Card)
- For the following peptide: group 3 C-NH, CH, H 4 group 1 HO. NH, group 4 N N-H group 2 At which pH will this structure remain stationary in an electric field? O A. when the pH is midway the pKas of groups 1 and 2 O B. when the pH is midway the pKas of groups 3 and 4 OC. when the pH is midway the pKas of groups 2 and 4 D. when the pH equals the pKa of group 1 OE. when the pH equals the pKa of group 4arrow_forwardIndicate which orbitals overlap to form the s bonds in each molecule.a. BeBr2 b. HgCl2 c. ICNarrow_forwardThis image shows us a of G6PD. A. monomeric (tertiary) structure B. homodimeric (quaternary) structure O C. heterodimeric (quaternary) structure O D. homotetrameric (quaternary) structure O E. heterotetrameric (quaternary) structurearrow_forward
- Match the chemical properties with the amino acid (note: the charges are not indicated on the charged amino acids) A. NH3+ - CH - COO- I (CH)2 I OH N,N - DimethyltryptamineAspartic acid B. NH3+ - CH - COO- I (CH)4 I NH2 Which one of each of these is: uncharged and polar charged and non-polar negatively charged and polar positively charged and polararrow_forwardDefine the following terms: a. hydrocarbon b. hydrophilic c. hydrophobic d. functional group e. R grouparrow_forwardDetermine the structure of the compound below. C10H1402 IR shows carbonyl and ether stretches. The compound is non-aromatic. 2H 24 2H 96 t heptet 1H 1. PPM 2 3H GH 6Harrow_forward
- Mestranol, sold under the brand names Enovid, Norinyl, and Ortho-Novum among others, is an estrogen medication which has been used in birth control pills. Select all of the functional group families, to which mestranol belongs. Choose one or more: O A. ester OB. alkene OC. ether O D. thiol O E. alcohol OF. arene OG. aldehyde OH. ketone O 1. alkyne OHarrow_forwardA sample was analyzed using mass spectrophotometer and molecular ion peak was obtained at m/z 120. Base peak was seen at m/z 91 and another peak of was observed at m/z 92. UV spectroscopic analysis revealed the presence of an aromatic ring. Determine the compound and justify your answer. And draw the mass spectrum.arrow_forwardTwo students plan and carry out an investigation to determine if ionic or covalent bonds have stronger intermolecular forces. They collect the following data. C6H1206 NaCI color white white texture,grain size rough, small pieces rough, very small grains solubility in water soluble (able to dissolve) soluble (able to dissolve) melting point melts easily does not melt a. Identify the type of intramolecular bonds that are involved. b. Which substance will have the stronger intermolecular forces? c. Which properties allow you to predict the strength of the intermolecular forces? Format B I U ... >arrow_forward
- Indicate whether each of the following statement about enantiomers is True or False. a. Enantiomers always have the same molecular formula. b. Enantiomers always have the same structural formula. c. Enantiomers are always nonsupetrimposable mirror images of each other. d. Enantiomers always differ in handedness.arrow_forwardA metal ion with a 2+ charge has 23 electrons and forms a compound with a halogen ion that contains 17 protons. a. What is the identity of the metal ion? b. What is the identity of the halogen ¡on and how many electrons does it contain? c. Determine the compound that it forms and name it.arrow_forwardWhich of the following statements is not true concerning the three molecules shown below? A В I. The three molecules are structural isomers. II. Structure C is properly named as a hexane. III. Structure B has the highest boiling point of the three molecules. IV. Structure A has the greatest induced dipole interactions of the three molecules.arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





