
Concept explainers
Interpretation: The product formed after the crossed-aldol condensation between 2-nitrobenzaldehyde and acetone in the first step for synthesis of indigo needs to be determined.
Concept Introduction:In crossed-aldol condensation, two different carbonyl compounds (both with alpha hydrogen atom) undergo condensation reaction together. There is a possibility of 4 products in such type of condensation reaction.
Explanation of Solution
The formation of indigo is represented as follows:
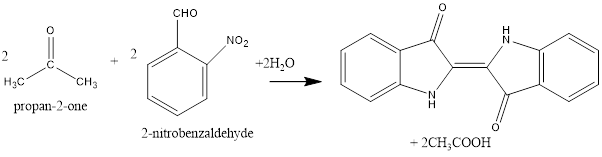
The first step of the reaction is crossed aldol condensation. The given reactants are 2-nitrobenzaldehyde and acetone or propan-2-one. Both of the reactantshasa carbonyl group thus, both are carbonyl compounds. Since, both the carbonyl compounds are different thus, crossed aldol condensation is possible.
The structure of reactants is represented as follows:
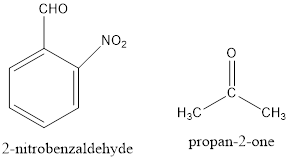
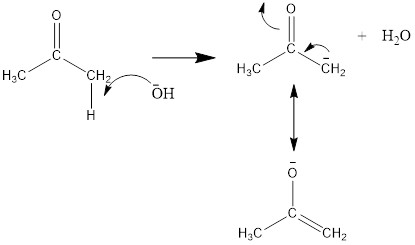
Here, the removal of alpha hydrogen from 2-nitrobenzaldehyde is not possible because the negative charge on the double bond is not stable. Thus, the hydroxide ion of water removes the alpha hydrogen of acetone or propan-2-one.
The mechanism for the crossed aldol condensation will be as follows:
Now, the negative charge will attack the carbonyl group of 2-nitrobenzaldehyde as follows and results in the formation of a hydroxy
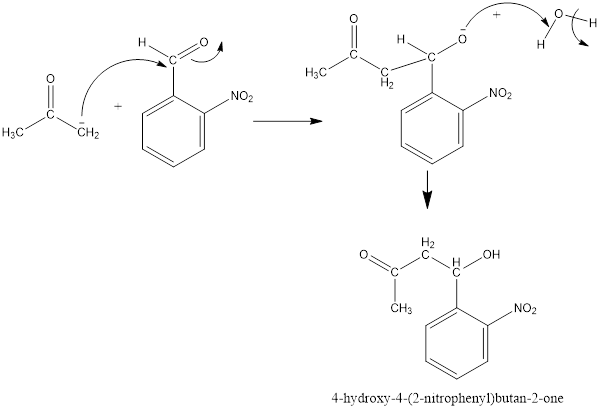
Thus, the product formed in the first step will be as follows:
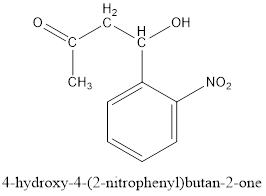
After this series of condensations, tautomerizations, a retro Claisen condensation and elimination reactions result in the formation of indigo.
Want to see more full solutions like this?
Chapter 45 Solutions
EBK A SMALL SCALE APPROACH TO ORGANIC L
- The compound 3,5,5-trimethyl-2-cyclohexenone can be synthesized using acetone and ethyl acetoacetate as sources of carbon atoms. New carbon-carbon bonds in this synthesis are formed by a combination of aldol reactions and Michael reactions. Show reagents and conditions by which this synthesis might be accomplished.arrow_forward-Hydroxyketones and -hydroxyaldehydes are also oxidized by treatment with periodic acid. It is not the -hydroxyketone or aldehyde, however, that undergoes reaction with periodic acid, but the hydrate formed by addition of water to the carbonyl group of the -hydroxyketone or aldehyde. Write a mechanism for the oxidation of this -hydroxyaldehyde by HIO4.arrow_forwardThe base-promoted rearrangement of an -haloketone to a carboxylic acid, known as the Favorskii rearrangement, is illustrated by the conversion of 2-chlorocyclohexanone to cyclopentanecarboxylic acid. It is proposed that NaOH first converts the a-haloketone to the substituted cyclopropanone shown in brackets and then to the sodium salt of cyclopentanecarboxylic acid. (a) Propose a mechanism for base-promoted conversion of 2-chlorocyclohexanone to the proposed intermediate. (b) Propose a mechanism for base-promoted conversion of the proposed intermediate to sodium cyclopentanecarboxylate.arrow_forward
- How does hcl react with this compound via acid-catalyzed electophilic aromatic substitution-alkylation?arrow_forwardWhat is the reaction mechanism of an acid catalyzed reaction converting 9-fluorenol to 9-methoxy fluorene.arrow_forwardhe reaction that occurs when the benzaldehyde in your hand is reacted in a basic environment. Cannizzaro reaction is called benzoin recovery when reacted with cyanide. It Explain the reason by writing the reaction mechanisms of the reactions.arrow_forward
- If in the Grignard synthesis of benzoic acid a student determined that his crude product was a mixture of benzoic acid and bromobenzene, map out a series of extractions using the following solvents and solutions to get pure benzene or its salt in an ether solution. Methyl tert-butyl ether (MTBE) 3M HCl 3M NaOHarrow_forwardThe following are intermediate products in the stepwise synthesis of compound 1 from benzene. Give the correct sequence of reactions with the appropriate reagents that will lead to the correct intermediate products and final product.arrow_forwardIf the following compound were treated with excess NaOH followed by an excess of D2O,what would be the structure of the new product formed?arrow_forward
- What factor seems to be responsible for the Wagner-Meerwein rearrangement of camphene to give isobornyl acetate (a 3º → 2º carbocation rearrangement)?arrow_forwardShow how the 2-phenylpropene acid can be synthesized from benzene:arrow_forward. Write short notes on the following, mentioning reagents, reaction conditions, products formed, mechanism, purification and precautions where necessary• Hydrolysis of 2-bromo-2-methylbutane• Nitration of benzene• Reduction of Nitrobenzene• Ammonolysis of benzoylchloride• Ozonolysis of 2-methylbut-2-enearrow_forward
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

