
Interpretation:
The minimum pressure at which the liquid water is the
Concept Introduction:
Phase:
A phase is defined as a homogeneous and physically distinct part of a system which is bounded by a surface and is mechanically separable from the other parts of the system.
Phase rule:
Phase rule is very important to predict the conditions that must be specified for a system to exhibit equilibrium. According to phase rule, for system in internal equilibrium,
Clausius Clapeyron equation:
On a pressure temperature diagram the line separating the two phases is known as the coexistence curve and the Clausius Clapeyron equation gives the slope of the tangents to this curve.
The equation is given as,
Where,
Phase diagram for water:
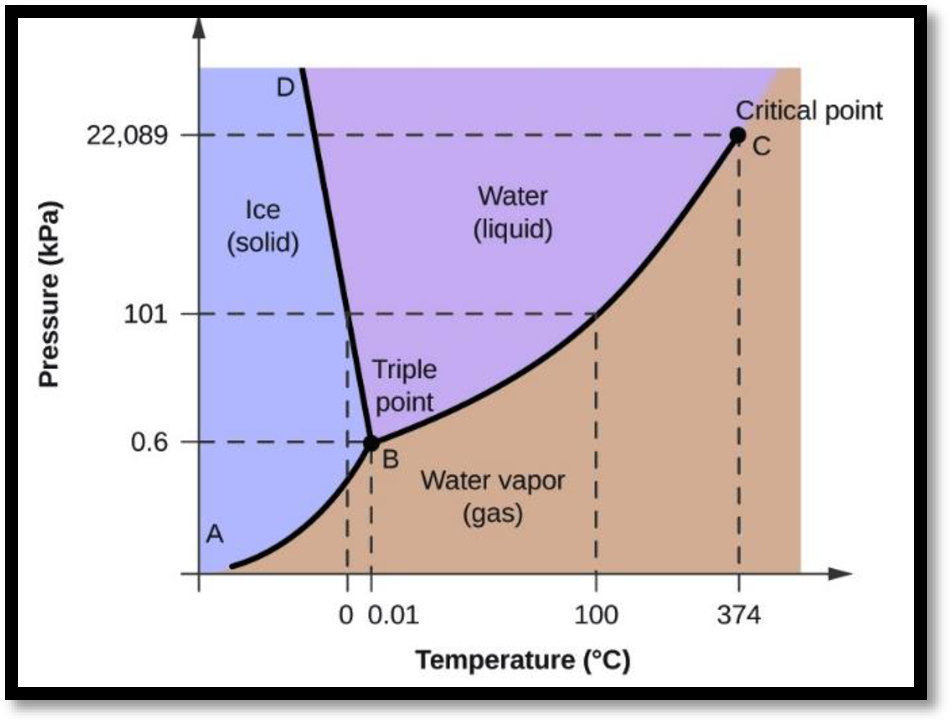
Figure-1
According to the figure BD represents the solid liquid equilibrium, BC represents the liquid vapor equilibrium and AB represents the solid vapor equilibrium.
Using Clausius Clapeyron equation from this phase diagram it can be predicted that, as the volume of the liquid water is less than the volume of ice so the slope for solid-liquid equilibrium boundary will be negative and as the difference is less so the value will be more. Thus it can be concluded that the melting point decreases with increase in pressure.
As the volume of vapor is more than volume of liquid hence the slope for the liquid vapor equilibrium will be positive but value of slope is less as the volume difference is more. Hence with increase in pressure the boiling point will increase.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Us Solutions Manual To Accompany Elements Of Physical Chemistry 7e
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





