
Concept explainers
Interpretation:
The labeled bonds in the given compound are to be arranged in the order of increasing bond lengths.
Concept introduction:
In hybridization, one
Answer to Problem 4.1P
The increasing order of bond-lengths in the given compound is
Explanation of Solution
The given compound is,
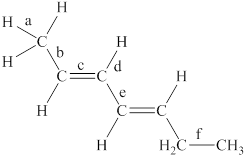
Figure 1
The bond with higher percentage of s-character has electron density closer to the nucleus and thus, has shorter bond length. The order of percentage
• Bond ‘a’ is present between the
• Bond ‘c’ is present between the two
• Due to the conjugation between two pi bonds, the bond length of ‘e’ is in between the carbon-carbon single bond and carbon-carbon double bond.
• Bond ‘b’ is present between one
• The
From the above points, it is concluded that the order of bond-lengths in the given compound is
The increasing order of bond-lengths in the given compound is
Want to see more full solutions like this?
Chapter 4 Solutions
EBK ORGANIC CHEMISTRY
- Curved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and follow the curved arrows to draw the resonance hybrid contributor. Include all lone pairs and charges as appropriate..arrow_forwardplease answer B regarding the question above.arrow_forwardCurved arrows are used to illustrate the flow of electrons. Using the provided resonance structures, draw the curved electron- pushing arrows to show the interconversion between resonance hybrid contributors. Be sure to account for all bond- breaking and bond-making stepsarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
