
Concept explainers
(a)
Show the direction
(a)
Explanation of Solution
Show the direction vector in the FCC unit cell for the cubic direction
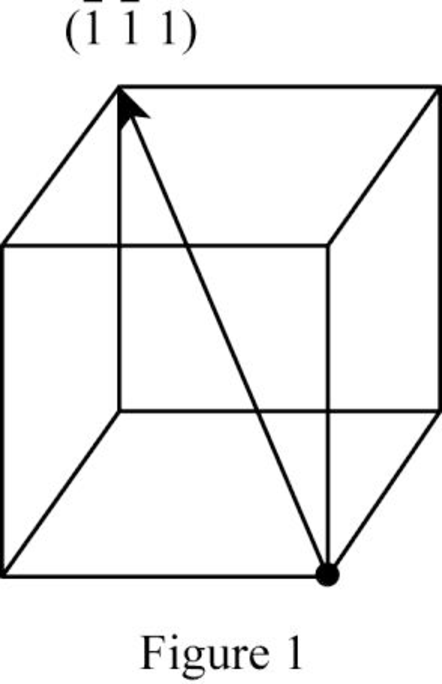
Position coordinates:
For the cubic direction
Repeat distance:
For the cubic direction
(b)
Show the direction vector in a FCC unit cell for the cubic direction
(b)
Explanation of Solution
Show the direction vector in the FCC unit cell for the cubic direction
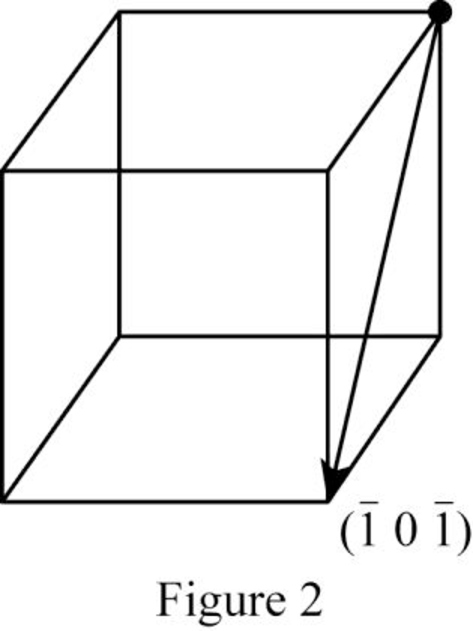
Position coordinates:
For the cubic direction
Repeat distance:
For the cubic direction
(c)
Show the direction vector in a FCC unit cell for the cubic direction
(c)
Explanation of Solution
Show the direction vector in the FCC unit cell for the cubic direction
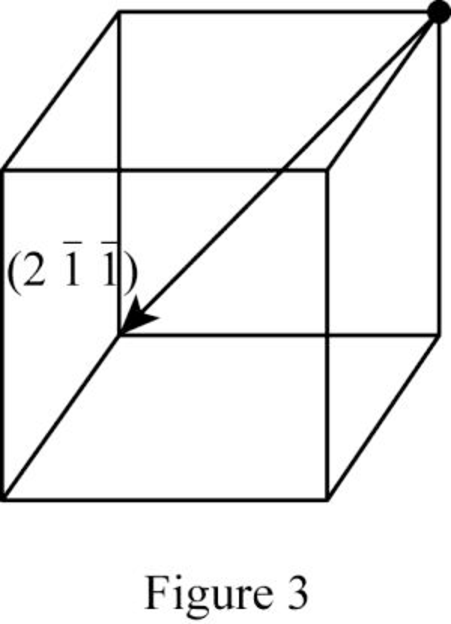
Position coordinates:
For the cubic direction
Repeat distance:
For the cubic direction
(d)
Show the direction vector in a FCC unit cell for the cubic direction
(d)
Explanation of Solution
Show the direction vector in the FCC unit cell for the cubic direction
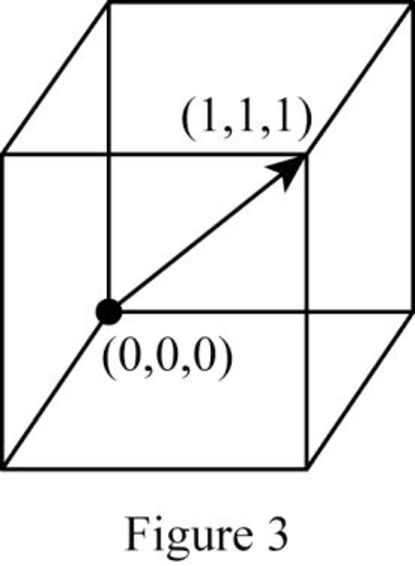
Position coordinates:
For the cubic direction
Repeat distance:
For the cubic direction
(e)
The angle between the direction vectors
(e)
Answer to Problem 30AAP
The angle between the direction vectors
Explanation of Solution
Write the expression to calculate angle between the direction vectors
Here, Miller indices of the cubic plane 1 are
Conclusion:
Substitute 1 for
Thus, the angle between the direction vectors
Want to see more full solutions like this?
Chapter 3 Solutions
Foundations of Materials Science and Engineering
- The density of BCC iron is 7.882g/cm3 and the lattice parameter is 0.2866nm when hydrogen atoms are introduced at interstitial positions. Calculate(a) the atomic fraction of hydrogen atoms; and(b) number of unit cells on average that contain hydrogen atoms.arrow_forwardDetermine the Miller indices for the planes of the cubic unit cell shown in figure 2, according to the coordinate axis system shown.arrow_forwardDetermine the repeat distance, linear density, and packing fraction for FCC nickel, which has a lattice parameter of 0.35167 nm, in the[100],[110] , and [111] directions. Which of these direction is close packed?arrow_forward
- Calculate the atomic densities of the directions [100], [111] and [110] in a face centered cubic structure.arrow_forwardConsider two hypothetical metallic crystal structures A and B. A has an fcc and B a bcc lattice structure. Both have the same unit cell volume. The Atomic Packing Factor for A is 0.740 and that for B is 0.680. What is the ratio of the fcc to bcc atomic radii? (This question has only one correct answer) a. 0.82 b. 0.63 c. 0.92 d. 0.44 e. 1.21 Clear my choicearrow_forwardIn a cubic crystal, draw any three (3) parallel planes and determine their miller indices.arrow_forward
- Beryllium with a crystalline structure HCP lattice constant c / a = 1.568 and the radius of the atom is 0.1143nm (calculate the volume of the unit cell and calculate the theoretical density if you know that the atomic weight A = 9.01g / mol)arrow_forwardDetermine the Miller indices of the cubic crystal plane that intersects the following position coordinates(points): (1, ½, 1), (½, 0, ¾), and (1, 0, ½). Sketch the plane, and show all steps and distances.arrow_forwardCalculate the gravitational constant g, in SI units, for alocation on the surface of the sun.arrow_forward
- qjly21 For FCC iron, calculate the diffraction angle for the (220) set of planes. The lattice parameter for Fe is 0.3571 nm. Assume that monochromatic radiation having a wavelength of 0.1540 nm is used, and the order of reflection is 1.arrow_forwardCalculate the volume of an FCC(Face Center Cubic) unit cell (cell edge length a) in terms of the atomic radiusarrow_forwardthe volume of BCC unit cell when the atomic radius is 0.662 nm isarrow_forward
 Understanding Motor ControlsMechanical EngineeringISBN:9781337798686Author:Stephen L. HermanPublisher:Delmar Cengage Learning
Understanding Motor ControlsMechanical EngineeringISBN:9781337798686Author:Stephen L. HermanPublisher:Delmar Cengage Learning
