
Interpretation: The shorthand structures of poly(vinyl chloride) and nylon
Concept introduction: The molecules which are obtained by the combination of similar units and possessing high molecular mass are known as
Answer to Problem 31.1P
The shorthand structures of poly(vinyl chloride) and nylon
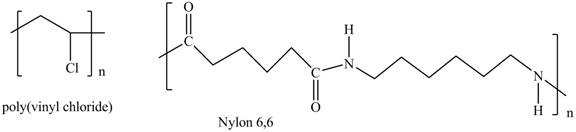
Explanation of Solution
The monomer required for the formation of poly(vinyl chloride) is vinyl chloride.
A shorthand structure of polymers represents the brackets that are placed around the repeating unit of the chain.
The shorthand structure of poly(vinyl chloride) and nylon
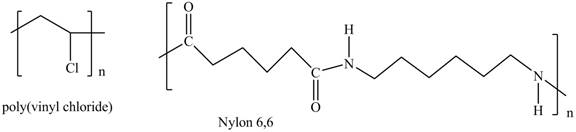
Figure 1
The shorthand structures of poly(vinyl chloride) and nylon
Want to see more full solutions like this?
Chapter 31 Solutions
Organic Chemistry
- Nylon can be destroyed easily by strong acids.Explain the chemical basis for the destruction.(Hint: The products are the starting materials of thepolymerization reaction.)arrow_forwardWhat does the part ‘6, 6’ mean in the name nylon-6, 6?arrow_forwardPolyethylene (PE), polyvinylchloride (PVC), polypropylene (PP), polystyrene, an nylon are examples ofarrow_forward
- Hexanedioic acid and 1,6-hexanediamine are two monomers that made up polymer Nylon 6,6. 5. Draw the structural formulae of both monomers. (a) Write a reaction equation between the monomers to form Nylon 6.6. (b) State the type of polymerisation. (c) Name the functional group in the polymer. (d) (e) Give the general name of this polymer. (f) Write the repeating unit of Nylon 6,6. Ahe structural formulae of the polymers formed from the polymerization of the followingarrow_forward1. What are biodegradable polymers and their classification ( include its functions and importance)?arrow_forwardWrite the repeating units expected from the following polymerization reaction(h)..arrow_forward
- Draw structural formulas for the repeat units in these polymers.(1) polymethylene(2) polyformaldehyde(3) poly(phenylene oxide)(4) poly(1-acetoxyethylene)(5) poly(methyl acrylate)arrow_forward1 In a laboratory a sample of nylon-6,10 was made by reacting an aqueous solution of 1,6-diaminohexane with decanedioyl chloride in a hexane solvent. a State the second product formed in this reaction. b Explain why an excess of 1,6-diaminohexane is used.arrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning





