
Chemistry
3rd Edition
ISBN: 9780073402734
Author: Julia Burdge
Publisher: MCGRAW-HILL HIGHER EDUCATION
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3, Problem 60QP
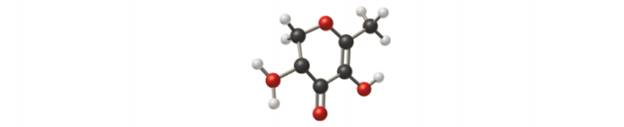
Researchers recently reported that the compound in stale beer that attracts cockroaches is DDMP, shown here.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Problem Draw a Lewis structure and identify the octet-rule exception for (a) H3PO4 (draw two resonance forms and select the more important); (b) BFCl2.Plan We draw each Lewis structure and examine it for exceptions to the octet rule.(a) The central atom is in Period 3, so it can have more than an octet.(b) The central atom is B, which can have fewer than an octet of electrons.
Match the following compounds.
which molecule is presented here ?
Chapter 3 Solutions
Chemistry
Ch. 3.1 - Practice ProblemATTEMPT Calculate the molecular or...Ch. 3.1 - Practice Problem BUILD
Calculate the molecular or...Ch. 3.1 - Practice ProblemCONCEPTUALIZE Some...Ch. 3.1 - What is the molecular mass of citric acid ( H 3 C...Ch. 3.1 - 3.1.2 What is the formula mass of calcium citrate...Ch. 3.2 - Practice Problem ATTEMPT
Determine the percent...Ch. 3.2 - Practice Problem BUILD
Determine the simplest...Ch. 3.2 - Practice Problem CONCEPTUALIZE
Determine the...Ch. 3.2 - 3.2.1 What is the percent composition by mass of...Ch. 3.2 - What is the percent composition by mass of sodium...
Ch. 3.3 - Prob. 1PPACh. 3.3 - Practice Problem BUILD
Write and balance the...Ch. 3.3 - Practice ProblemCONCEPTUALIZE Write a balanced...Ch. 3.3 - What are the stoichiometric coefficients in the...Ch. 3.3 - 3.3.2 Which chemical equation represents the...Ch. 3.3 - 3.3.3 Which is the correctly balanced form of the...Ch. 3.3 - 3.3.4 Carbon monoxide reacts with oxygen to...Ch. 3.4 - Prob. 1PPACh. 3.4 - Practice Problem BUILD
Write and balance the...Ch. 3.4 - Prob. 1PPCCh. 3.4 - How many molecules are in 30 .1 g of sulfur...Ch. 3.4 - How many moles of hydrogen are there in 6 .50 g of...Ch. 3.4 - 3.4.3 Determine the empirical formula of a...Ch. 3.4 - Determine the empirical formula of a compound that...Ch. 3.5 - Practice ProblemATTEMPT Potassium is the second...Ch. 3.5 - Practice Problem BUILD
Calculate (a) the number...Ch. 3.5 - Practice Problem CONCEPTUALIZE
These diagrams show...Ch. 3.5 - What is the empirical formula of a compound...Ch. 3.5 - 3.5.2 What are the empirical and molecular...Ch. 3.5 - 3.5.3 Determine the masses of and produced by...Ch. 3.5 - How is it possible for the combined masses of CO 2...Ch. 3.6 - Prob. 1PPACh. 3.6 - Practice Problem BUILD
(a) Determine the mass of...Ch. 3.6 - Practice ProblemCONCEPTUALIZE Plain doughnuts from...Ch. 3.6 - 3.6.1 How many moles of will be produced if Li...Ch. 3.6 - 3.6.2 Determine the stoichiometric amount (in...Ch. 3.7 - Prob. 1PPACh. 3.7 - Practice ProblemBUILD (a) Determine the number of...Ch. 3.7 - Practice ProblemCONCEPTUALIZE A particular...Ch. 3.7 - 3.7.1 What mass of is produced according to the...Ch. 3.7 - 3.7.2 What is the percent yield for a process in...Ch. 3.7 - How many moles of NH 3 can be produced by the...Ch. 3.7 - What mass of water is produced by the reaction of...Ch. 3.7 - Reactants A (red) and B (blue) combine to form a...Ch. 3.7 - Which of the following represents the contents of...Ch. 3.8 - Practice ProblemATTEMPT Determine the empirical...Ch. 3.8 - Practice ProblemBUILD Determine the empirical...Ch. 3.8 - Practice ProblemCONCEPTUALIZE What is the smallest...Ch. 3.9 - Practice ProblemATTEMPT The combustion of a 28...Ch. 3.9 - Practice ProblemBUILD Determine the mass of CO 2...Ch. 3.9 - Prob. 1PPCCh. 3.10 - Prob. 1PPACh. 3.10 - Prob. 1PPBCh. 3.10 - Prob. 1PPCCh. 3.11 - Practice ProblemATTEMPT Calculate the mass of...Ch. 3.11 - Practice Problem BUILD
What mass of glucose must...Ch. 3.11 - Prob. 1PPCCh. 3.12 - Prob. 1PPACh. 3.12 - Prob. 1PPBCh. 3.12 - Practice ProblemCONCEPTUALIZE The diagrams show a...Ch. 3.13 - Prob. 1PPACh. 3.13 - Practice Problem BUILD
What mass of ether will be...Ch. 3.13 - Prob. 1PPCCh. 3.14 - Prob. 1PPACh. 3.14 - Practice Problem BUILD
Using the chemical species...Ch. 3.14 - Prob. 1PPCCh. 3 - 3.1
Calculate the mass of water produced in the...Ch. 3 - 3.2
How much can be produced?
(a)
350.0...Ch. 3 - 3.3
How much can be produced?
(a)
91.51...Ch. 3 - 3.4
How much of the excess reactant remains when...Ch. 3 - What is meant by the term molecular mass, and why...Ch. 3 - Explain the difference between the terms molecular...Ch. 3 - Calculate the molecular mass (in amu) of each of...Ch. 3 - Calculate the molecular mass (in amu) of each of...Ch. 3 - Calculate the molecular mass or formula mass (in...Ch. 3 - 3.6 Calculate the molecular mass or formula mass...Ch. 3 - 3.7 Use ammonia to explain what is meant by the...Ch. 3 - 3.8 Describe how the knowledge of the percent...Ch. 3 - Tin (Sn) exists in Earth's crust as SnO 2 ....Ch. 3 - 3.10 For many years, chloroform was used as an...Ch. 3 - All the substances listed here are fertilizers...Ch. 3 - Limonene. shown here, is a by-product of the...Ch. 3 - 3.13 Tooth enamel is . Calculate the percent...Ch. 3 - A four-pack of Red Bull Energy Drink consists of...Ch. 3 - 3.15 A “variety pack" of ramen noodles consists of...Ch. 3 - Prob. 16QPCh. 3 - Prob. 17QPCh. 3 - 3.18 Why must a chemical equation be balanced?...Ch. 3 - Prob. 19QPCh. 3 - Write an unbalanced equation to represent each of...Ch. 3 - Write an unbalanced equation to represent each of...Ch. 3 - Prob. 22QPCh. 3 - For each of the following unbalanced chemical...Ch. 3 - Prob. 24QPCh. 3 - Prob. 25QPCh. 3 - Which of the following equations best represents...Ch. 3 - Prob. 27QPCh. 3 - Define the term mole. What is the unit for mole in...Ch. 3 - Prob. 29QPCh. 3 - Prob. 30QPCh. 3 - If we know the empirical formula of a compound,...Ch. 3 - Prob. 32QPCh. 3 - The thickness of a piece of paper is 0.0036 in....Ch. 3 - Prob. 34QPCh. 3 - Prob. 35QPCh. 3 - Prob. 36QPCh. 3 - How many grams of gold ( Au ) are there in 15.3...Ch. 3 - Prob. 38QPCh. 3 - Prob. 39QPCh. 3 - 3.40 What is the mass in grams of lead atoms?
Ch. 3 - Prob. 41QPCh. 3 - Prob. 42QPCh. 3 - Which of the following has a greater mass: two...Ch. 3 - Prob. 44QPCh. 3 - Prob. 45QPCh. 3 - Prob. 46QPCh. 3 - Prob. 47QPCh. 3 - Prob. 48QPCh. 3 - Prob. 49QPCh. 3 - Prob. 50QPCh. 3 - Prob. 51QPCh. 3 - Prob. 52QPCh. 3 - Prob. 53QPCh. 3 - Prob. 54QPCh. 3 - Prob. 55QPCh. 3 - Prob. 56QPCh. 3 - Prob. 57QPCh. 3 - Prob. 58QPCh. 3 - In response to invasion by a microorganism, the...Ch. 3 - Researchers recently reported that the compound in...Ch. 3 - Prob. 61QPCh. 3 - Prob. 62QPCh. 3 - 3.63 Menthol is a flavoring agent extracted from...Ch. 3 - 3.64 Ascorbic acid (vitamin C) contains C, H, and...Ch. 3 - The amino acid cysteine plays an important role in...Ch. 3 - 3.66 The diagram shows the products of a...Ch. 3 - 3.67 Which of the following diagrams could...Ch. 3 - 3.68 On what law is stoichiometry based? Why is it...Ch. 3 - Prob. 69QPCh. 3 - Prob. 70QPCh. 3 - Prob. 71QPCh. 3 - Prob. 72QPCh. 3 - Prob. 73QPCh. 3 - Prob. 74QPCh. 3 - 3.75 When baking soda (sodium bicarbonate or...Ch. 3 - Prob. 76QPCh. 3 - Prob. 77QPCh. 3 - Prob. 78QPCh. 3 - Prob. 79QPCh. 3 - Prob. 80QPCh. 3 - Prob. 81QPCh. 3 - Prob. 82QPCh. 3 - Prob. 83QPCh. 3 - Prob. 84QPCh. 3 - Prob. 85QPCh. 3 - Prob. 86QPCh. 3 - Prob. 87QPCh. 3 - Prob. 88QPCh. 3 - Prob. 89QPCh. 3 - Prob. 90QPCh. 3 - Nitroglycerin ( C 3 H 5 N 3 O 9 ) is a powerful...Ch. 3 - Prob. 92QPCh. 3 - Prob. 93QPCh. 3 - Prob. 94QPCh. 3 - Prob. 95QPCh. 3 - Prob. 96QPCh. 3 - Prob. 97QPCh. 3 - Prob. 98QPCh. 3 - Consider the reaction N 2 +3H 2 → 2NH 3 Assuming...Ch. 3 - 3.104 Determine whether each of the following...Ch. 3 - Prob. 101QPCh. 3 - Prob. 102APCh. 3 - Prob. 103APCh. 3 - Prob. 104APCh. 3 - Prob. 105APCh. 3 - Prob. 106APCh. 3 - Prob. 107APCh. 3 - 3.112 The carat is the unit of mass used by...Ch. 3 - An iron bar weighed 664 g. After the bar had been...Ch. 3 - Prob. 110APCh. 3 - Suppose you are given a cube made of magnesium...Ch. 3 - Prob. 112APCh. 3 - Prob. 113APCh. 3 - Prob. 114APCh. 3 - Calculate the number of cations and anions in each...Ch. 3 - Prob. 116APCh. 3 - 3.121 Avogadro’s number has sometimes been...Ch. 3 - Prob. 118APCh. 3 - In the formation of carbon monoxide. CO, it is...Ch. 3 - Prob. 120APCh. 3 - Prob. 121APCh. 3 - A compound made up of C, H, and Cl contains 55.0...Ch. 3 - Prob. 123APCh. 3 - Prob. 124APCh. 3 - Prob. 125APCh. 3 - Prob. 126APCh. 3 - Prob. 127APCh. 3 - 3.132 A mixture of methane and ethane of mass...Ch. 3 - Prob. 129APCh. 3 - A die has an edge length of 1.5 cm. (a) What is...Ch. 3 - Prob. 131APCh. 3 - Prob. 132APCh. 3 - A sample containing NaCl, Na 2 SO 4 , and NaNO 3...Ch. 3 - Prob. 134APCh. 3 - Prob. 135APCh. 3 - Prob. 136APCh. 3 - An impure sample of zinc (Zn) is treated with an...Ch. 3 - One of the reactions that occurs in a blast...Ch. 3 - Prob. 139APCh. 3 - Prob. 140APCh. 3 - Prob. 141APCh. 3 - 3.146 Aspirin or acetylsalicylic acid is...Ch. 3 - Prob. 143APCh. 3 - Prob. 144APCh. 3 - Lysine, an essential amino acid in the human body,...Ch. 3 - Prob. 146APCh. 3 - Prob. 147APCh. 3 - Prob. 148APCh. 3 - Prob. 149APCh. 3 - Cysteine, shown here, is one of the 20 amino acids...Ch. 3 - Prob. 151APCh. 3 - Carbon dioxide ( CO 2 ) is the gas that is mainly...Ch. 3 - Prob. 153APCh. 3 - Prob. 154APCh. 3 - Prob. 155APCh. 3 - Prob. 156APCh. 3 - 3.161 Potash is any potassium mineral that is used...Ch. 3 - Prob. 158APCh. 3 - Prob. 159APCh. 3 - Prob. 160APCh. 3 - Prob. 1SEPPCh. 3 - Prob. 2SEPPCh. 3 - Prob. 3SEPPCh. 3 - Prob. 4SEPP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- SOlve the next problem by showing step by step on a picture please. explain too.arrow_forwardI need help drawing the VSEPR geometry of ( CH2O), the electronic, molecular geometry, and if it’s polar. All of these get me confuse. Please helparrow_forwardGive typed full explanation Look at figure 3-22 that shows the electron density that occurs abound the Si-O bond. This electron density map gives the "shape" of the O and Si atoms when they are bonded together. Think about the answer in Q9 and choose the best response below: (Select answer choice) a. This figure shows that the Si and O atoms, when they bond together, do not form spheres, which is due to the fact that the Si-O bond is strongly covalent and these shared electrons affect atomic shape. This change in shape limits the applicability of Pauling's Coordination principle since that principle is based on the geometry of perfect spheres. b. This figure shows that the Si and O atoms, when they bond together are close to perfect spheres, which is due to the fact that the Si-O bond is strongly covalent. This figure shows that Pauling's Coordination principle should apply very precisely to any substance that contains Si-O bonds c. This figure shows that the Si and O atoms, form in a…arrow_forward
- Which elements on the periodic table (other than H) are likely to form a+1 cation?arrow_forward2b) NH₂ NH₂arrow_forwardAnswer this question: Based on the results of the solubility tests and chemical tests, what is the most probable structure of C6H5C3H5O? Draw your answer in the box You may draw the compound usingline-bond formula OR Lewis structure. Problem statement:You were tasked to propose a structure for a compound with a chemical formula of C6H5C3H5O. In the laboratory, the compound appears as a clear, colorless liquid. See table 1, for the results of solubility tests for C6H5C3H5O. Table 1. Results of the solubility tests for C6H5C3H5O. SOLUBILITY TEST OBSERVATIONS +(soluble/miscible)/ - (insoluble) distilled water two layers of liquid - 10% NaHCO3 two layers of liquid - 10% NaOH two layers of liquid - 10% HCl two layers of liquid - concentrated H2SO4 pinkish solution + Based on the results of the solubility tests, the compound can be grouped in VI, which is insoluble in water, 10% NaOH and 10% HCl but soluble in concentrated H2SO4. If the…arrow_forward
- For each of the following molecules, complete the following. (Please Explain) Draw the bond dipole for each polar bond. Indicate whether there is a net dipole or not using the blanks provided. You don’t need to provide the direction of the net dipole, only whether or not one exists.arrow_forwardDoxorubicin, shown here, is an important chemotherapy drug used to treat avariety of cancers, including bladder cancer, breast cancer, and certain forms of leukemia. Doxorubicin works by binding to DNA in such a way that a portion of it penetrates the DNA double helix— a process called intercalation. During transcription— the process that forms RNA— portions of the DNA strands are temporarily separated for the base sequence to be read and then are reconnected. With bound doxorubicin, however, the double helix does not reform properly after the strands are separated, which disrupts replication— the process that forms an identical copy of DNA. Which portion of doxorubicin do you think intercalates into the DNA double helix, and why do you think it has little difficulty doing so?arrow_forwardSupply a molecular formula structure for the compound shown herearrow_forward
- SOLVE THE FOLLOWING PROBLEM AND SHOW YOUR COMPLETE AND DETAILED SOLUTIONS FOR BETTER UNDERSTANDING.arrow_forwardKCl Express your answer as an ion.arrow_forwardConsider the Lewis structure shown for thionitromethane. Draw the major resonance structure for the compound shown; include lone pairs of electrons, formal charges, and condensed hydrogen atoms (located in the More menu). Then draw curved arrows to show how this can be converted to the Lewis structure given.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Types of bonds; Author: Edspira;https://www.youtube.com/watch?v=Jj0V01Arebk;License: Standard YouTube License, CC-BY