
(a)
Interpretation:
Concept Introduction:
Atoms are made up of even smaller particles. These particles are very small and these are all the building blocks of atoms and they are known as subatomic particles. Protons, electrons, and neutrons are the subatomic particles that are found in atom. Electrons possess a negative electrical charge. Protons possess a positive electrical charge. Neutrons possess no charge and they are neutral.
Mass number is the sum of the number of protons and neutrons inside the nucleus of an atom. This gives the number of subatomic particle present inside the nucleus. Mass number is represented by the symbol A.
From atomic number and mass number, the number of each sub atomic particle can be found.
Complete chemical symbol notation can be given as.
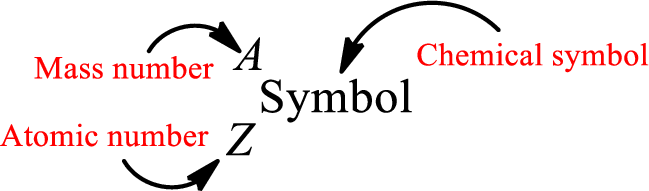
An element is a pure substance that cannot be broken by ordinary
(a)
Explanation of Solution
For
The atomic number is given as 7. The element is found to be nitrogen. Mass number given for nitrogen is given as 13. The number of protons present in it is also 7 as atomic number is the number of protons or electrons. Mass number of the sum of protons and neutrons present in the atom.
The number of neutrons can be identified by finding the difference between mass number and atomic number. This gives the number of neutrons to be 6.
The total number of nucleons is same as that of the mass number. This means the total number of nucleons is 13.
The total number of subatomic particle present in the given atom is the sum of mass number and atomic number. This means the total number of subatomic particle is 20.
For
The atomic number is given as 6. The element is found to be carbon. Mass number given for carbon is given as 13. The number of protons present in it is also 6 as atomic number is the number of protons or electrons. Mass number of the sum of protons and neutrons present in the atom.
The number of neutrons can be identified by finding the difference between mass number and atomic number. This gives the number of neutrons to be 7.
The total number of nucleons is same as that of the mass number. This means the total number of nucleons is 13.
The total number of subatomic particle present in the given atom is the sum of mass number and atomic number. This means the total number of subatomic particle is 19.
On comparing both the pair of atoms it is found that they contain same number of nucleons only.
(b)
Interpretation:
Concept Introduction:
Atoms are made up of even smaller particles. These particles are very small and these are all the building blocks of atoms and they are known as subatomic particles. Protons, electrons, and neutrons are the subatomic particles that are found in atom. Electrons possess a negative electrical charge. Protons possess a positive electrical charge. Neutrons possess no charge and they are neutral.
Atomic number for each and every element is a unique one. This is the total number of protons that is present in an atom. As the atom is electrically neutral, it can also be said that the total number of electrons is the atomic number. Atomic number is represented by the symbol Z.
Mass number is the sum of the number of protons and neutrons inside the nucleus of an atom. This gives the number of subatomic particle present inside the nucleus. Mass number is represented by the symbol A.
From atomic number and mass number, the number of each sub atomic particle can be found.
Complete chemical symbol notation can be given as.
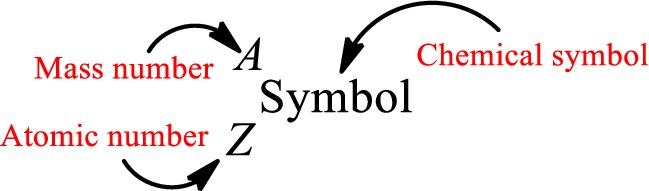
An element is a pure substance that cannot be broken by ordinary chemical reactions into simpler substances. All the atoms in an element will have the same atomic number. The electrons only take part in the chemical reaction while the nucleus does not. Hence, the atomic number (number or protons) does not change and it characterizes an atom.
(b)
Explanation of Solution
For
The atomic number is given as 17. The element is found to be chlorine. Mass number given for chlorine is given as 37. The number of protons present in it is also 17 as atomic number is the number of protons or electrons. Mass number of the sum of protons and neutrons present in the atom.
The number of neutrons can be identified by finding the difference between mass number and atomic number. This gives the number of neutrons to be 20.
The total number of nucleons is same as that of the mass number. This means the total number of nucleons is 37.
The total number of subatomic particle present in the given atom is the sum of mass number and atomic number. This means the total number of subatomic particle is 54.
For
The atomic number is given as 18. The element is found to be argon. Mass number given for argon is given as 36. The number of protons present in it is also 18 as atomic number is the number of protons or electrons. Mass number of the sum of protons and neutrons present in the atom.
The number of neutrons can be identified by finding the difference between mass number and atomic number. This gives the number of neutrons to be 18.
The total number of nucleons is same as that of the mass number. This means the total number of nucleons is 36.
The total number of subatomic particle present in the given atom is the sum of mass number and atomic number. This means the total number of subatomic particle is 54.
On comparing both the pair of atoms it is found that they contain same number of subatomic particles in them.
(c)
Interpretation:
Concept Introduction:
Atoms are made up of even smaller particles. These particles are very small and these are all the building blocks of atoms and they are known as subatomic particles. Protons, electrons, and neutrons are the subatomic particles that are found in atom. Electrons possess a negative electrical charge. Protons possess a positive electrical charge. Neutrons possess no charge and they are neutral.
Atomic number for each and every element is a unique one. This is the total number of protons that is present in an atom. As the atom is electrically neutral, it can also be said that the total number of electrons is the atomic number. Atomic number is represented by the symbol Z.
Mass number is the sum of the number of protons and neutrons inside the nucleus of an atom. This gives the number of subatomic particle present inside the nucleus. Mass number is represented by the symbol A.
From atomic number and mass number, the number of each sub atomic particle can be found.
Complete chemical symbol notation can be given as.
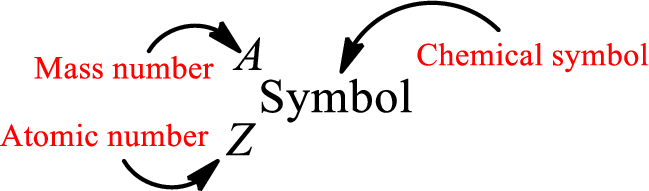
An element is a pure substance that cannot be broken by ordinary chemical reactions into simpler substances. All the atoms in an element will have the same atomic number. The electrons only take part in the chemical reaction while the nucleus does not. Hence, the atomic number (number or protons) does not change and it characterizes an atom.
(c)
Explanation of Solution
For
The atomic number is given as 17. The element is found to be chlorine. Mass number given for chlorine is given as 35. The number of protons present in it is also 17 as atomic number is the number of protons or electrons. Mass number of the sum of protons and neutrons present in the atom.
The number of neutrons can be identified by finding the difference between mass number and atomic number. This gives the number of neutrons to be 18.
The total number of nucleons is same as that of the mass number. This means the total number of nucleons is 35.
The total number of subatomic particle present in the given atom is the sum of mass number and atomic number. This means the total number of subatomic particle is 52.
For
The atomic number is given as 17. The element is found to be chlorine. Mass number given for chlorine is given as 37. The number of protons present in it is also 17 as atomic number is the number of protons or electrons. Mass number of the sum of protons and neutrons present in the atom.
The number of neutrons can be identified by finding the difference between mass number and atomic number. This gives the number of neutrons to be 20.
The total number of nucleons is same as that of the mass number. This means the total number of nucleons is 37.
The total number of subatomic particle present in the given atom is the sum of mass number and atomic number. This means the total number of subatomic particle is 54.
On comparing both the pair of atoms it is found that they contain same number of protons.
(d)
Interpretation:
Concept Introduction:
Atoms are made up of even smaller particles. These particles are very small and these are all the building blocks of atoms and they are known as subatomic particles. Protons, electrons, and neutrons are the subatomic particles that are found in atom. Electrons possess a negative electrical charge. Protons possess a positive electrical charge. Neutrons possess no charge and they are neutral.
Atomic number for each and every element is a unique one. This is the total number of protons that is present in an atom. As the atom is electrically neutral, it can also be said that the total number of electrons is the atomic number. Atomic number is represented by the symbol Z.
Mass number is the sum of the number of protons and neutrons inside the nucleus of an atom. This gives the number of subatomic particle present inside the nucleus. Mass number is represented by the symbol A.
From atomic number and mass number, the number of each sub atomic particle can be found.
Complete chemical symbol notation can be given as.
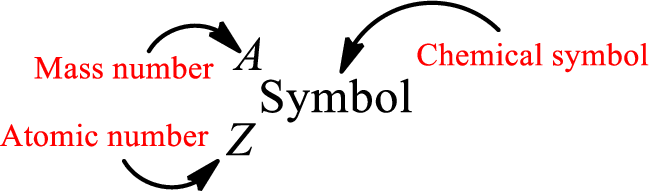
An element is a pure substance that cannot be broken by ordinary chemical reactions into simpler substances. All the atoms in an element will have the same atomic number. The electrons only take part in the chemical reaction while the nucleus does not. Hence, the atomic number (number or protons) does not change and it characterizes an atom.
(d)
Explanation of Solution
For
The atomic number is given as 8. The element is found to be oxygen. Mass number given for oxygen is given as 18. The number of protons present in it is also 8 as atomic number is the number of protons or electrons. Mass number of the sum of protons and neutrons present in the atom.
The number of neutrons can be identified by finding the difference between mass number and atomic number. This gives the number of neutrons to be 10.
The total number of nucleons is same as that of the mass number. This means the total number of nucleons is 18.
The total number of subatomic particle present in the given atom is the sum of mass number and atomic number. This means the total number of subatomic particle is 26.
For
The atomic number is given as 9. The element is found to be fluorine. Mass number given for fluorine is given as 19. The number of protons present in it is also 17 as atomic number is the number of protons or electrons. Mass number of the sum of protons and neutrons present in the atom.
The number of neutrons can be identified by finding the difference between mass number and atomic number. This gives the number of neutrons to be 10.
The total number of nucleons is same as that of the mass number. This means the total number of nucleons is 19.
The total number of subatomic particle present in the given atom is the sum of mass number and atomic number. This means the total number of subatomic particle is 28.
On comparing both the pair of atoms it is found that they contain same number of neutrons.
Want to see more full solutions like this?
Chapter 3 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- An isotope of sodium (Na) has a mass number of 22. How many neutrons does it have? a. 11 b. 12 c. 22 d. 44arrow_forwardThe atomic number gives thea. number of neutrons in the nucleus.b. number of protons in the nucleus.c. weight of the atom.d. number of protons in the valence shell.arrow_forwardA carbon atom contains six protons and six neutrons.A. What are its atomic number and atomic weight?B. How many electrons does it have?C. How many additional electrons must it add to fill its outermost shell? How does this affect carbon’s chemical behavior?D. Carbon with an atomic weight of 14 is radioactive. How does it differ in structure from nonradioactive carbon? How does this difference affect its chemical behavior?arrow_forward
- The name given to the atomic particle found outside the nucleus of an atom isa. proton.b. neutron.c. electron.d. ion.arrow_forwardLabel the following as a protein, nucleic acid, carbohydrate, or lipid. DNA 11. 12. Sugar 13. Fats 14. Starch 15. RNA 16. Hair 17. Oils 18. Fingernails Use the periodic table to answer questions 19-26. 19. What is the atomic number of chromium? 12 a. b, 24 c. 52 What is the mass number of chromium? 20. a. 12 b. 24 с. 52 How many neutrons are there in chromium? a. 28 21. b. 24 c. 12 22. _How many protons are in sodium? а. 12 b. 11 C. 24 23. How many electrons does fluorine have? a. 17 b. 9 C. 19 24. How many neutrons does aluminum have? a. 17 b. 14 С. 18 Which element has 48 electrons? 25. a. Titanium b. Cadmium C. Barium How many neutrons does sodium have? 26. a. 23 b. 12 С. 11arrow_forwardIsotopes differ from each other only in the number of electrons they contain. Select one: a. True b. Falsearrow_forward
- Which of the following levels in the shell model of an atom can hold up to two electrons? A. The second shell B. The first shell C. The third shellarrow_forward6. You are investigating the contents of three weird aliens. You want to see which biomolecules the aliens are made of. Based on the percentages of elements the aliens have, which biomolecules does your alien have. Please explain your answer as well. Alien 1: 33% C, 40% O, 4% H, 14% N, and 9% P Alien 2:25% C, 25% O, 50% H, 0%N, and 0%P Alien 3: 25% C, 25% O, 48%H, 2% N, and 0% P Alien 1: Alien 2: Alien 3:arrow_forwardWhich of the following contain copper atoms (Cu*) Select one: Complex II b. Complex III O C. Complex IV Od. Complex Iarrow_forward
- Atoms are incredibly tiny, so small that most microscopes can't even see them, but atoms themselves are made of even smaller particles: protons, neutrons, and electrons. Which of the following describes where protons are located in an atom? A. A shell B. The nucleus C. Both the nucleus and a shellarrow_forwardSome prokaryotic and all eukaryotic cells use oxygen to harvest energy from food molecules. In what form is that energy available to power cell work? a) b) c) glucose molecules d) fat molecules ATP molecules heat and light P Sunlight energy ECOSYSTEM Photosynthesis in chloroplasts Cellular respiration in mitochondria ATP (for cellular work) ww www Heat energyarrow_forwardSmoke detectors in homes and apartments contain Am-241, which undergoes alpha decay. What is the abbreviated symbol of the "daughter nuclide" produced in the nuclear reaction? Hint: atomic number of Am is 95. A. a particle B. Pu-237 C. Am-237 D. He - 4 Е. Np-237arrow_forward

 Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College

