
Concept explainers
(a)
Interpretation:
The structure for the product of each of the given Lewis acid-base association reactions is to be derived by using curved-arrow notation. The formal charge on each species is to be assigned. The Lewis acid and the Lewis base is to be labeled. The atom that donates electrons in each case is to be predicted.
Concept introduction:
Lewis acid-base association reaction involves the transfer of a proton from a base to an acid. Lewis acid accepts an electron pair and acts as an electrophile. Lewis base donates an electron pair and acts as a nucleophile.
Answer to Problem 3.1P
The Lewis acid and Lewis base are
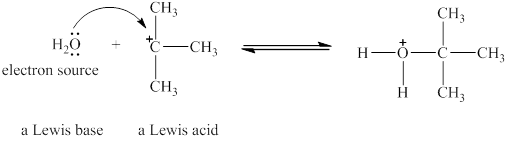
Explanation of Solution
Lewis acids are electron donor, whereas Lewis bases are electron pair accepter. In water molecule oxygen consists of a lone pair. Thus, it will act a Lewis base and oxygen atom has tendnecy to donate its electrons. The
The oxygen in
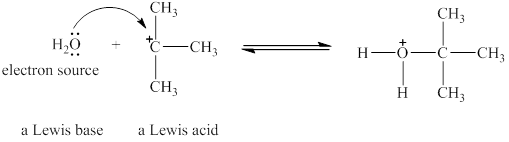
Figure 1
The Lewis acid and Lewis base are
(b)
Interpretation:
The structure for the product of each of the given Lewis acid-base association reactions is to be derived by using curved-arrow notation. The formal charge on each species is to be assigned. The Lewis acid and the Lewis base is to be labeled. The atom that donates electrons in each case is to be predicted.
Concept introduction:
Lewis acid-base association reaction involves the transfer of a proton from a base to an acid. Lewis acid accepts an electron pair. It acts as an electrophile Lewis base donates an electron pair. It acts as nucleophile.
Answer to Problem 3.1P
The Lewis acid and Lewis base are
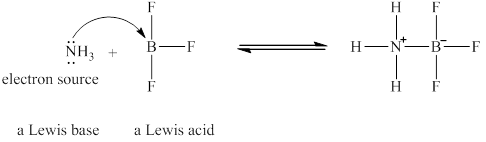
Explanation of Solution
Lewis acids are electron donor, whereas Lewis bases are electron accepter. In
The nitrogen in
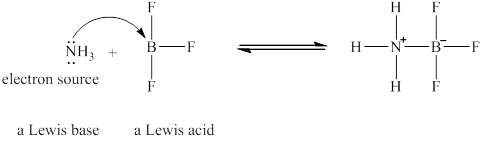
Figure 2
The Lewis acid and Lewis base are
Want to see more full solutions like this?
Chapter 3 Solutions
EBK ORGANIC CHEMISTRY
- Arrange in increasing acidity. Then explain the answer in terms of their partial charge and inductive effectsarrow_forwardIf pH of solution is 13, it means that if is:(a) Weakly acidic(b) Weakly basic(c) Strongly acidic(d) Strongly Basicarrow_forwardExplain using examples how electronic factors (resonance and inductive effects) can be used to determine acidity and basicity of organic compounds. Use electronic and stability factors to account for the acidity of each highlighted hydrogen in the molecule below. Identify the most acidic proton. (a) HO. (b) HO. (c) H.arrow_forward
- For each conjugate acid-base pair, identify the first species as an acid or a base and the second species as its conjugate acid or base. In addition, draw Lewis structures for each species, showing all valence electrons and any formal charge. (a) CH3CH3 CH3CH2-arrow_forwardThe structural formulas of pseudoephedrine (a free base) and pseudoephedrine hydrochloride (a quaternary ammonium compound) are shown below. Which of these two compounds would you predict to be more water-soluble? Can you explain to me and help me, please?arrow_forwardPlease provide a synthesis of the following question - please be as detailed as posisble in the explanation.arrow_forward
- (a) .H (b)arrow_forwardProvide the bond line structures of two acids, one whose conjugate base is resonance stabilized and one which is not. Explain the effect that resonance will have on acidity.arrow_forwardExplain what will happen to the (i) yield and (ii) purity, of aspirin if the following steps were modified in its synthesis Modification in Step Yield Explanation Changing from phosphoric acid to sulfuric acid.arrow_forward
- Identify the most acidic compound between the following moleculesDraw the resonance structures of the conjugate base of the acid chosen in (a). Use the curved arrows to show the movement of electrons and the resonance arrows to show the relationship between these structures. All lone electron pairs and charges must be clearly indicatedarrow_forwardHi , I have the problem to answer quetion 2.51 (b) and (d).. May I have the scheme draft and explanation to understand this topic better.. Can you explain to me how we need to choose the factor that determine acid strength based on the question given , as i know there are four factors included (element effect,inductive effect,resonance effect and hybridization effect).arrow_forwardGive a clear handwritten answer with explanation..given below a reaction label the acid ,conjugate acid, base ,andconjugate base..and tell the side of equilibrium is favored then show the electron pushing arrows to reflect the mechanism of actionarrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


