
General, Organic, and Biological Chemistry (3rd Edition)
3rd Edition
ISBN: 9780134042428
Author: Laura D. Frost, S. Todd Deal
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3, Problem 3.103AP
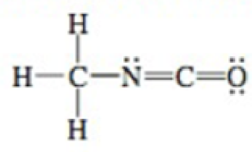
Methyl isocyanate is used in the manufacturing of pesticides and was the deadly gas released from a chemical plant in the 1984 disaster in Bhopal, India, that killed thousands. Determine the shape around each of the carbon in the Lewis structure of methyl isocyanate.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Explain the difference between Ionic and Covalent Bonds.
Draw the Lewis structures of the neutral atom and the ion of
Carbon. Determine the number of electrons gained or lost in forming an ion.
Explain the differences among nonpolar covalent bonds, polar covalent bonds, and ionic bonds.
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw Lewis structures showing all valence electrons for these molecules. (a) C2H6 (b) CS2 (c) HCNarrow_forwardplease draw a lewis structure, determine the # of electron groups around the central atom, and determine the geometry of the molecule. (please state both the name of the geometry and draw the molecular shape) molecule lewis structure electron pair geometry around the central atom(s) molecular shape around the central atom(s) CO2 H2S H2O2 NF3 SO2Cl2 NO2 C2H2 C2H4 C2H6arrow_forwardExplain the difference between a polar and a nonpolarcovalent bond.arrow_forward
- Draw the Lewis structure of these compounds and use the electronegativity values, calculate the polarity of the molecules and write if the compounds will be polar, nonpolar, or ionic. Label the + and -next to the atoms of the molecule if they have charge. Also, write the overall dipole. Cl₂ HCl NH₃ CH₂O CH₄ O₂ CH₂Cl₂ H₂O CH₃Li HCN CH₂CHCl CH₃CH₃ CO₂arrow_forwardWhich of the following statements could be true regarding polar molecules?Choose one or more: A polar molecule will not contain polar bonds. A polar molecule may have one or more lone pairs. A polar molecule has an uneven distribution of electron density. A polar molecule will never contain nonpolar bonds. The bond dipoles in a polar molecule will cancel.arrow_forwardDraw a Lewis structure for the given skeletal molecule. Note: Multiple bonds and lone pairs are not shown in the skeletal molecule. H H H H H N O 0-C N-C-C-ċ-OH C-C нн H-O H How many lone pairs of electrons are found in the Lewis structure of your molecule?arrow_forward
- what is the difference between Ionic and covalent bonds?arrow_forwardUse the Molecular Polarity Simulation to complete the following. Effect of Molecular Geometry on Polarity For each molecule: Sketch each molecule as shown in the simulation. Include arrows to show the bond dipoles as well as a molecular dipole (if present). Circle polar or nonpolar to indicate the polarity of the molecule. Name the molecular geometry (from Part A) H2O vs CO2 H2O CO2 polar nonpolar polar nonpolar Molecular Geometry Molecular Geometry Question 1a: How does the molecular geometry (linear vs bent) affect the molecular polarity?arrow_forwardConsider the resonance structures of formate. Select the true statements about the resonance structures. Each carbon-oxygen bond is somewhere between a single and double bond. The actual structure of formate is an average of the two resonance forms. Each oxygen atom has a double bond 50% of the time. The actual structure of formate switches back and forth between the two resonance forms.arrow_forward
- Draw Lewis structures for each molecular formula. C2H4Cl2 (two isomers) C3H8O (three isomers)arrow_forwardConsider the resonance structures of formate. Y-Y > H. Select the true statements about the resonance structures. Each carbon-oxygen bond is somewhere between a single and double bond. Each oxygen atom has a double bond 50% of the time. The actual structure of formate is an average of the two resonance forms. The actual structure of formate switches back and forth between the two resonance forms.arrow_forwardThe compound XeCl2F2 can exist in two different forms. One form is polar and the other form is non-polar. Draw a valid Lewis structure of XeCl2F2. Then, draw two 3-dimensional representations of this molecule (including all lone pairs); one that shows the polar form with a correct dipole arrow and the other that shows the non-polar form of the molecule.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co
Types of bonds; Author: Edspira;https://www.youtube.com/watch?v=Jj0V01Arebk;License: Standard YouTube License, CC-BY