
In 1976, a team of chemists in the United Kingdom was developing new insecticides by modifying sugars with chlorine (Cl2), phosgene (Cl2CO), and other toxic gases. One young Member of the team misunderstood his verbal instructions to “test” a newly made candidate substance. He thought he had been told to “taste” it. Luckily for him, the substance was not toxic, but it was very sweet. It became the food additive sucralose.
Sucralose has three chlorine atoms substituted for three hydroxyl groups of sucrose (table sugar). It binds so strongly to the sweet-taste receptors on the tongue that the human brain perceives it as 600 times sweeter than sucrose. Sucralose was originally marketed as an artificial sweetener called Splenda”, but it is now available under several other brand names.
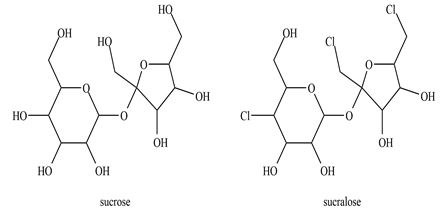
Researchers investigated whether the body recognizes sucralose as a carbohydrate. They began by feeding sucralose labeled with 14C to volunteers. Analysis of the radioactive molecules in the volunteers’ urine and feces showed that 92.8 percent of the sucralose passed through the body without being altered. Many people are worried that the chlorine atoms impart toxicity to sucralose. How would you respond to that concern?
Trending nowThis is a popular solution!

Chapter 3 Solutions
BIOLOGY:CONCEPTS+APPL.(LOOSELEAF)
- How do you synthesize Chloramphenicol and show the mechanism? Explain step by step please. (If you can 1 sentence is enough.) (Drug Chemistry)arrow_forwardPlace numbers beside the statements below to indicate the order of events occurring in the light reactions. I know for sure that 1 and 8 are correct, but the rest I’m not too confident about. I attached both my work and a blank copy.arrow_forwardWhich of the following statements is FALSE? A. saccharin is 300 times sweeter than saccharose B. D-glucose is slightly salty and L-glucose is sweet. C. a-D-mannose is sweet and B-D-mannose is bitter D. The stereochemistry of flavor-producing agents plays an important role in the specific sensation of flavor.arrow_forward
- Anthocyanins are another type of plant pigment that are purple or red. Say you run a chromatography experiment using the same setup as you did in this lab, and you find that anthocyanins do not move from the line of origin. Assuming the solvent traveled the same distance as in your experiment (D in Table 1 above), what is the Rf value of anthocyanin?arrow_forwardI want clear handwritten solution of both parts....i will up votearrow_forwardMargarines made from plants oils are healthier ,since they are hydrogenated. A) TRUE B)FALSEarrow_forward
- Please draw the Pharmaceutical label(Manufacturer lebel) and highlight the parts ,drawing should be handwritten i include a example, answer should be like this.......arrow_forwardPenicillin is an irreversible inhibitor. What is the name of the enzyme that it inhibits? How does that make penicillin a useful drug for us?arrow_forwardSulfamethoxazole is a type of sulfonamide antibiotic. Its absorbance ranges from 250-300 nm in a UV spectrophotometer, which tells that it is in the UV region. What characteristic/s of sulfamethoxazole is responsible for the absorbance in the UV region? Please answer in detail and clearly. Thank you!arrow_forward
- A researcher wishes to obtain a glycoside (an organic compound) from an aqueous plant extract using the solvent extraction technique. She therefore performs the following steps: Step 1: An aqueous solution (100 mL) containing 7 g of the glycoside was shaken with 50 mL of ethyl acetate. Step 2: The resulting aqueous layer from Step 1 was further shaken with 50 mL of dichloromethane. Given that: Kp for the glycoside in ethyl acetate-water = 0.8 Kp for the glycoside in dichloromethane-water = 0.9 Calculate the total mass of glycoside extracted. If the molar mass of the glycoside = 784.3 g/mol, calculate the molar concentration of the glycoside remaining in the aqueous layer after each extraction.arrow_forwardName the three basic classes of drug targets in medicinal chemistry and indicate, which are the most/least important as targets for biologics? I don't sure if I answer these questions correctly. Hope someone can give me confirmation and explain to me how enzymes will be affected by bioloigcs? My answer so far: The three common classes of drug targets are; receptors, nucleic acid, and enzymes. As a target for biologics, the least important will be a nucleic acid, and the most important will be receptor and second enzymes. Biologic drugs such as monoclonal antibodies will bind to the antigens, which are overexpressed receptor that need to be deactivated. In this case, the monoclonal antibodies will act as an antagonist and block the ligand from binding to the antigens, which are extensively presented in cancer cells. In addition, it can also affect enzymes by.......?arrow_forwardWhy does acetazolamide makebeer taste flat?arrow_forward
