
Concept explainers
Interpretation:
The curved arrows are to be added to the given reactions, which indicate the flow of electrons for all the bond-forming and bond-breaking steps.
Concept introduction:
Curved arrows are used to represent the direction of the flow of electrons in a reaction mechanism.
Curved arrows are drawn in such a way that they point from the source of an electron pair toward the atom, which receives the electron pair.
The direction of flow of electrons is always from a high electron density site to a low electron density site.
Curved arrows never represent the movement of atoms.
The movement of electrons shown by the curved arrows should not violate the octet rule for the elements, which are present in the second row of the periodic table.
Electrophiles are positive or partially positive species that attract electrons or negatively charged species toward itself.
Answer to Problem 1PP
Solution:
(a)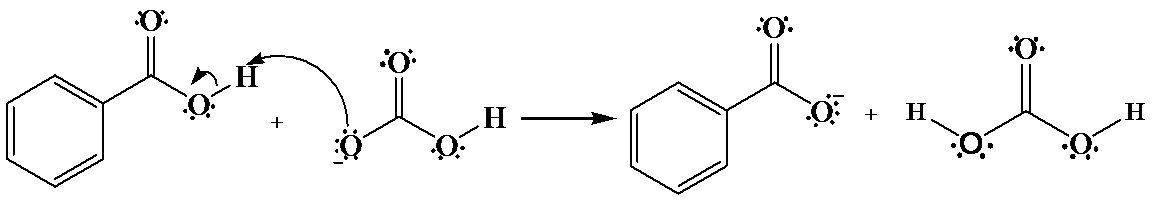
(b) 
Explanation of Solution
a)

The hydrogen of benzoic acid is partially positive and is electrophilic in nature. An oxygen atom provides an unshared pair of electrons, which forms bond with the benzoic acid hydrogen that cause the departure of a benzoate anion.
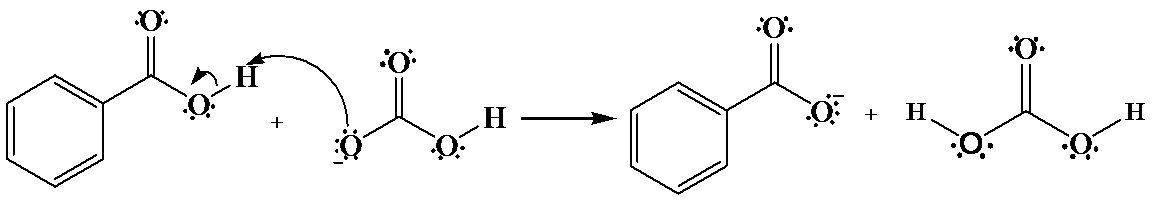
b)
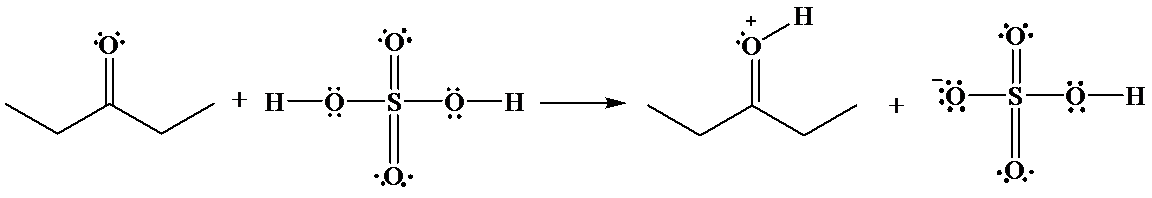
The hydrogen of sulfuric acid is partially positive and is electrophilic in nature. An oxygen atom of
The reaction is as follows:

Want to see more full solutions like this?
Chapter 3 Solutions
Organic Chemistry, 12e Study Guide/Student Solutions Manual
- Follow the format of solving the problem where you should write the GIVEN, ASKED, SOLUTION, and ANSWER. Add curved arrows to the following reactions to indicate the flow of electrons for all of the bond-forming and bond-breaking steps.arrow_forwardplease help with all parts (a-c). I do not understandarrow_forwardConsider the reaction. Draw the FULL electron pushing mechanism for the reaction including ALL intermediates (with lone pairs and formal charges), explicitly depicting any hydride shifts, and draw ALL electron pushing arrows.arrow_forward
- For each of the following, write the major product(s) and then draw out each step in the mechanism using curved arrows. Show ALL lone pair electrons and formal charges. Redraw ALL molecules as to show explicitly ALL bonds being broken or formed. Identify the molecular orbital (HOMO) of the nucleophile and the molecular orbital (LUMO) of electrophile involved in the nucleophilic attack. MO diagrams are not necessary. (a). (b). -CHO 2. H₂O 1. LiAlH4 2. H₂O MgBr , etherarrow_forwardOrganic Chemistry Loudon | Parise SEVENTH EDITION Draw the major organic product for each of the two situations. (a) Phenylacetic acid is treated first with Br₂ and one equivalent of PBr3, then with a large excess of ethanol. Be on Incorrect (b) Propionic acid is treated first with Br, and one equivalent of PBr3, then with a large excess of ammonia. presented by Macmillan Leaming Br f NH,arrow_forwardProvide a complete curved arrow mechanism that accounts for the transformation below. Be sure to show all arrows, reation intermediates, lone pairs, and formal chargesarrow_forward
- Give a clear handwritten answer..complete the following reaction..choose the perfect arrow for given reactionarrow_forward(a) Identify the missing major products A and B from the reactions below. ( b) For the catalyst used for each transformation, draw the structure and state the formal electron count and oxidation state (showing your workings)arrow_forwardPlease answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. Long and detailed explanations are not needed. Keep it short, brief, and direct because I only need the answers. If you mix equal amounts of compounds B and C, the resulting mixture is?arrow_forward
- The curved-arrow convention depicts the flow of electrons, including bond-forming and breaking events. Draw the outcome of the following reaction based on the provided curved arrow. Be vigilant regarding formal charges. :ci: B. Ci: Draw three additional resonance structures of acetamide (below) and use curved arrow notation to show how the resonance structures are formed. Label the resonance contributors alphabetically (ex. A, B, C, D) and rank them from most to least significant contributor. •oº• Ⅱ. H3C 0°• H N Harrow_forward1)Predict the product and 2)using curved arrows to show electron movement, provide the reasonable mechanism for the reactionarrow_forward[References] Draw the expected major organic product of the Sharpless epoxidation of each allylic alcohol using (-)-diethyl tartrate as the chiral catalyst. (a) (b) All hydrogen atoms are implied. If a chiral atom is attached to a hydrogen atom, you should not show the hydrogen atom but use either a wedge or a dashed bond. Apply formal charges where appropriate. Omit lone pairs and radical electrons from your answer. • Omit + signs between structures. ● HO O HO -OCH3 کر ? ChemDoodle Previous Nextarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





