
Concept explainers
(a)
Interpretation: Primary, secondary and tertiary hydrogen in below structure should be labeled.
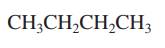
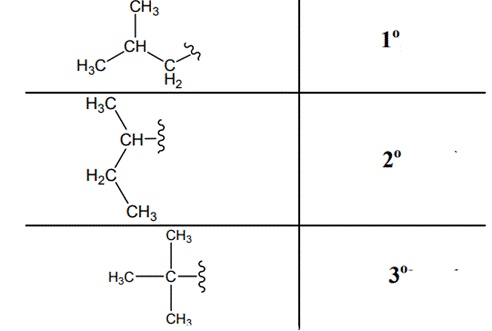
Concept introduction: Primary, secondary and tertiary hydrogen are identified as indicated below:
(a)
Explanation of Solution
The carbon linked to one alkyl/carbon while other two
The carbon linked to two alkyl/carbons and one
(b)
Interpretation: Primary, secondary and tertiary hydrogen in below structure should be labeled.
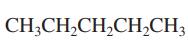
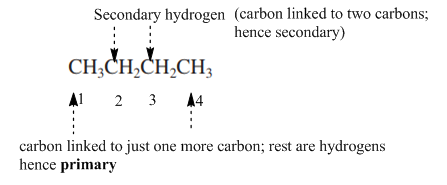
Concept introduction: Primary, secondary and tertiary hydrogen are identified as indicated below:
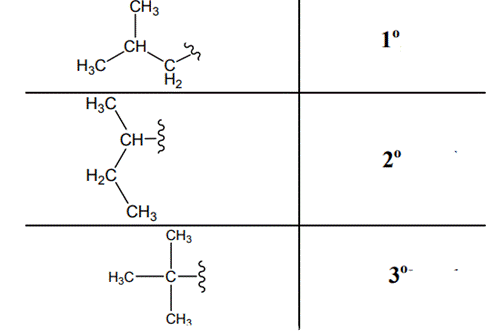
(b)
Explanation of Solution
The carbon linked to one alkyl/carbon while other two
The carbon linked to two alkyl/carbons and one
(c)
Interpretation: Primary, secondary and tertiary hydrogen in below structure should be labeled.
Concept introduction: Primary, secondary and tertiary hydrogen are identified as indicated below:
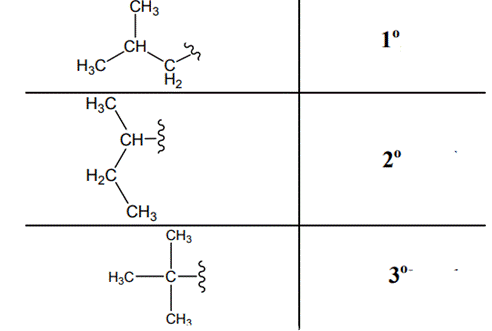
(c)
Explanation of Solution
The carbon linked to one alkyl/carbon while other two
The carbon linked to two alkyl/carbons and one

(d)
Interpretation: Primary, secondary and tertiary hydrogen in below structure should be labeled.
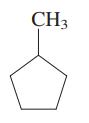
Concept introduction: The carbon linked to one alkyl/carbon while other two
The carbon linked to two alkyl/carbons and one
These are indicated below:
(d)
Explanation of Solution
Since each of the carbon of ring is linked to each other they represent secondary; the methyl group attached to ring represents primary as it is directly linked to only one carbon that is ring carbon. Thus it denotes primary hydrogens. Thus three types of hydrogens are identified and labeled below.
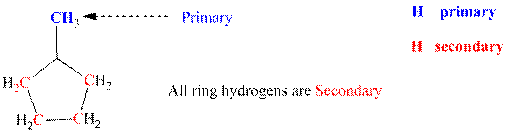
Want to see more full solutions like this?
Chapter 3 Solutions
EBK ORGANIC CHEMISTRY
- What is C terminal amino adid of the peptide below? HO N. HO H2N N. N. Select one: a. H,N-CH- O H,N-CH-C OHarrow_forwardCH2 H3C H3C COOH Reagents e. PB13 f. NaCN then H3O* g. NBS, CCI4 h. CrO3, H2SO4 а. HBr b. Mg, ether c. CO2, ether then H3O* d. BH3, THF then H2O2, OH"arrow_forwardCl Br h. -CH3 Br T H₂C-CH i. CH Cl CH₂CH3arrow_forward
- Choose the correct structure for each of the following compounds. a. an alcohol with molecular formula C₂H6O O CH3 CH₂-O-CH3 O CH3-C-H OCH3-CH₂-OH O CH3-O-CH3 b. an aldehyde with molecular formula C3H$O ů OCH, CCH, O CH3-CH₂-O-CH3 OCH3 CH₂-CH OH CH3-CH-CH3arrow_forwardReagents a. CeHsCHO b. NaOH, ethanol c. Pyrrolidine, cat. H* d. H2C=CHCN e. H30* f. LDA g. ELOC(=0)CO,Et h. BRCH2CH=CH2 Na* OEt, ethanol j. Br2, H* k. K* t-BuO 1. CH2(CO2Et)2 m. heat Select reagents from the table to synthesize this compound from cyclopentanone. Enter the letters of the chosen reagents, in the order that you wish to use them, without spaces or punctuation (i.e. geda). Submit Answer Try Another Version 1 Item attempt remainingarrow_forwardO CH3-C-O-CH3 O || CH3-CH₂-C-OH C-OH 1. NaOH 2. H3O+ 1. SOCI₂ 2. CH3NH₂ CH3OH H+ cat.arrow_forward
- 3. Predict the major products of the following reactions. Include stereochemistry where appropriate.arrow_forward3.* CH, CH, CH3 HO HSO, CH,-CH-CH-CH, CH,-C-CH, CH, CH,-C CH-CH3 HO. HO GHI IHG GHI IHGarrow_forward17. Which of the following structures are constitutional isomers? CH3 CH3 CH3 CH3 H3C II III IV O A.I, II, & II B. I, II, & IV C.I& II D. All are constitutional isomers E. All are configurational isomers 18. What is the IUPAC name of the compound shown below? CH3 CH(CH3)2 H,C H. A. 1,2,3-Trimethylbutane B. 2,3-Dimethylpentane C. 2,3,4-Trimethylpentane D. 2-1sopropylbutane OE 22,3-Trimethylbutane O O OO0arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





