
Tutorials In Introductory Physics: Homework
1st Edition
ISBN: 9780130662453
Author: Lillian C. McDermott, Peter S. Shaffer
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 27.1, Problem 2bTH
A long pin is used to hold the piston in place as shown in the diagram. The cylinder is then placed into boiling water.
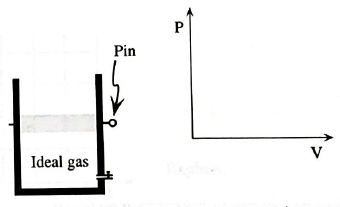
b. Sketch this process in the PV diagram at right.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Referring to the figure shown, water contained in a piston–cylinder assembly, initially at 100 kPa and 25°C. The water is heated until the final temperature reaches to 320°C. Kinetic and potential energy effects are negligible.Sketch the process on T-v diagram.
Determinea. The quality of the mixture when piston hits the stop.b. The final pressure.Hint: Establish the problem in three states; 1: initial, 2: when piston hits the stop, 3: at T3 = 320°C.
0.0040 mol of gas undergoes the process as shown.a. What type of process is this?b. What are the initial and final temperatures?
An ideal gas undergoes two thermodynamic processes as shown in
FIGURE 1.
a. Name the processes AB and BC.
b. If the initial temperature is 40°C, find the temperatures at B and C.
c. Find the total work done
d. What is the total change in internal energy for these processes
Chapter 27 Solutions
Tutorials In Introductory Physics: Homework
Ch. 27.1 - Prob. 1aTHCh. 27.1 - In this process, which of the quantities P, V, n,...Ch. 27.1 - Consider the following incorrect student...Ch. 27.1 - Explain why it is not possible to use the ideal...Ch. 27.1 - A long pin is used to hold the piston in place as...Ch. 27.1 - A long pin is used to hold the piston in place as...Ch. 27.1 - Prob. 2cTHCh. 27.2 - Prob. 1aTHCh. 27.2 - Prob. 1bTHCh. 27.2 - Prob. 1cTH
Additional Science Textbook Solutions
Find more solutions based on key concepts
6. A 12 V DC power supply is connected to the primary coil of a transformer. The primary coil has 100 turns and...
College Physics: A Strategic Approach (4th Edition)
The pV-diagram of the Carnot cycle.
Sears And Zemansky's University Physics With Modern Physics
26. Is the period longer or shorter for orbits of greater altitude?
Conceptual Physical Science (6th Edition)
5. A rocket firing its engine and accelerating in outer space (no gravity, no air resistance) suddenly runs out...
College Physics (10th Edition)
(a) An 8.00/F capacitor is connected in parallel to another capacitor, producing a total capacitance of 5.00/F ...
University Physics Volume 2
57. (I) How long will It take a 2750-W motor to lift a 385-kg piano to a sixth-story window 16.0 m above?
Physics: Principles with Applications
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- Instruction: Answer the problems completely. Show your complete solution. Write legibly. A cylinder contains one mole of oxygen gas at a temperature of 27°C. The cylinder is provided with a frictionless piston, which maintains a constant pressure at 1 atm on the gas. The gas heated until its temperature increases at 127°C. a. Draw a diagram representing the process in the pV-plane.arrow_forwardPlease help me with d, e, farrow_forwardSolve the problemarrow_forward
- How much thermal energy needs to be extracted for the temperature to drop to 3x10^2 K? USE IMAGE AS REFERNCEarrow_forwardFirst Law of Thermodynamics PROCEDURES: in the pictures attached A.When gas expands, Is the (W) work done by the system or (W) work done on the system? Explain your answer. B.Compare the temperature outside (surroundings) and inside (system) the bottle before pouring the boiling water into the bucket. C.Compare the temperature outside (surroundings) and inside (system) the bottle after pouring the boiling water into the bucket.arrow_forwardThe figure(Figure 1) shows the thermal energy of 0.15 mol of gas as a function of temperature. Part A What is Cy for this gas? Express your answer as an integer and include the appropriate units. HẢ ? Cy = Value Units Submit Figure <) 1 of 1 Eth (J) 1892 1492 1092 T (°C) 100 200arrow_forward
- Please show complete solutions with derivation of formulasarrow_forwardAnswer is also mentioned in picture containing the question. Kindly check the answer before submitting The solution.arrow_forwardShown below are bar charts for processes involving ideal gasses. For each, draw the appropriate line or curve on the PV graph, including an arrow for direction. Explain. 1. Temperature Pressure Volume bul IF IF I F 2. Temperature Pressure Volume T I FI FI F 4. I F IF I F P Temperature Pressure Volume 7 G I FI FI F P 3. For this, use the ideal gas law as a guide: Temperature Pressure Volume P V P V Varrow_forward
- First Law of Thermodynamics PROCEDURES: in the pictures attached A.When gas expands, Is the (W) work done by the system or (W) work done on the system? Explain your answer. B.Compare the temperature outside (surroundings) and inside (system) the bottle before pouring the boiling water into the bucket. C.Compare the temperature outside (surroundings) and inside (system) the bottle after pouring the boiling water into the bucket.arrow_forwardA gas with an initial temperature of 900°C undergoes the process as shown.a. What type of process is this?b. What is the final temperature?c. How many moles of gas are there?arrow_forwardOne cubic meter of oxygen at initial pressure of 1 bar and a temperature of 17°C are compressed isentropically to a volume of 0.50m 3. Use the specific heat at constant volume and pressure are 0.919 kJ/kgk and 0.659 kJ/kgK, respectively. determine the: a. The thermodynamic process the problem represents b. the pressure, temperature, and volume after the process c. change in internal energy, change in enthalpy and change in entropy d. work nonflow and heat transferredarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning


College Physics
Physics
ISBN:9781938168000
Author:Paul Peter Urone, Roger Hinrichs
Publisher:OpenStax College

Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning
Thermodynamics: Crash Course Physics #23; Author: Crash Course;https://www.youtube.com/watch?v=4i1MUWJoI0U;License: Standard YouTube License, CC-BY