
(a)
Interpretation: The starting materials that are needed to synthesize the given compound by a thermal
Concept introduction: A
Answer to Problem 27.40P
The starting materials that are needed to synthesize the given compound by a thermal
Explanation of Solution
The given product is shown below.
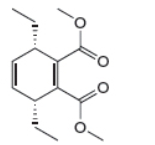
Figure 1
Retro synthesis can be carried out to identify the diene and dienophile for the given product.
The reaction that shows the disconnection approach of the given product is shown below.
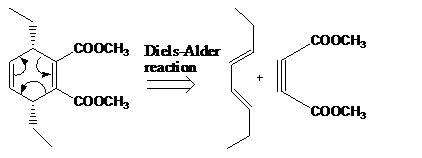
Figure 2
In the given product, the ring is opened due to the rearrangement of
The starting materials that are needed to synthesize the given compound by a thermal
(b)
Interpretation: The starting materials that are needed to synthesize the given compound by a thermal
Concept introduction: A chemical reaction that involves
Answer to Problem 27.40P
The starting materials that are needed to synthesize the given compound by a thermal
Explanation of Solution
The given product is shown below.
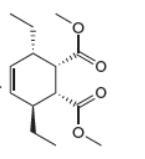
Figure 3
Retro synthesis can be carried out to identify the diene and dienophile for the given product.
The reaction that shows the disconnection approach of the given product is shown below.
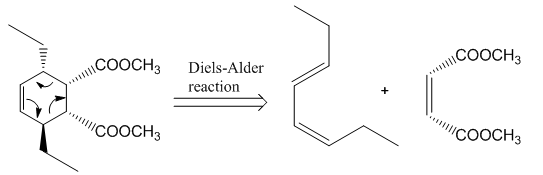
Figure 4
In the given product, the ring is opened due to the rearrangement of
Thus, the starting materials that are needed to synthesize the given compound by a thermal
The starting materials that are needed to synthesize the given compound by a thermal
(c)
Interpretation: The starting materials that are needed to synthesize the given compound by a thermal
Concept introduction: A chemical reaction that involves
Answer to Problem 27.40P
The starting materials that are needed to synthesize the given compound by a thermal
Explanation of Solution
The given product is shown below.
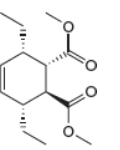
Figure 5
Retro synthesis can be carried out to identify the diene and dienophile for the given product.
The reaction that shows the disconnection approach of the given product is shown below.
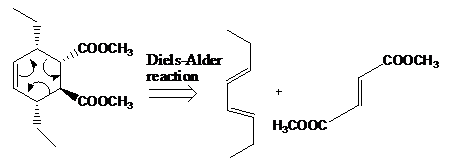
Figure 6
In the given product, the ring is opened due to the rearrangement of
Thus, the starting materials that are needed to synthesize the given compound by a thermal
The starting materials that are needed to synthesize the given compound by a thermal
Want to see more full solutions like this?
Chapter 27 Solutions
Organic Chemistry
- Draw the major organic products for the reaction of 1,4-dimethylcyclohexa-1-3-diene with catalytic HBr in H2O.arrow_forwardThe bicyclic alkene P can be prepared by thermal electrocyclic ringclosure from cyclodecadiene Q or by photochemical electrocyclic ringclosure from cyclodecadiene R. Draw the structures of Q and R, andindicate the stereochemistry of the process by which each reactionoccurs.arrow_forwardDraw the cycloalkene that would react with the reagent given to account for the product formed. ? + H₂O H₂SO4 CH3 ▼ • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. » [ ]# CH3 ? OHarrow_forward
- Synthesize each compound from (CH3)3CH.arrow_forwardDraw the product formed from cyclohex-2-enone in each of three reactions. Reaction 1 Reaction 2 Reaction 1 1. NaCN 1. (CH3)2CULI Reaction 2 product product 2. H30*, H2O 2. CH3CH2I 1. EtO,CCH2CO½ET NaOEt, HOET 2. Hао", Н-о, д Reaction 3 Reaction 3 productarrow_forwardThe electrophilic addition of HBr to 3-cyclohexylbut-1-ene gives a mixture of two constitutional isomers. These two isomers can also be produced from 3-cylcohexybut-2-ene, but one of which requires different reaction conditions; the electrophilic addition of HBr to 3-cylcohexybut-2-ene produces one of these isomers while the electrophilic addition of HBr to 3-cylcohexybut-2-ene, in the presence of peroxides, produces the other one. Draw the structure for the isomer product that can result from the reactants 3-cyclohexylbut-1-ene and 3-cylcohexybut-2-ene using different reaction conditions. Part 1 of 2 Click and drag to start drawing a structure. C 8 DY 000 8: >arrow_forward
- What are the starting reactants for the following reaction? 4 X = cyclohexa-1,3-diene; Y = ethyl acrylate X = cyclopenta-1,3-diene; Y = acrylaldehyde X = cyclohexa-1,3-ene; Y = ethyl acrylate X = hexa-1,3-diene; Y = ethyl propiolate CO₂C₂H5arrow_forwardDraw the alkene that would react with the reagent given to account for the product formed. ? + H₂O H₂SO4 CH3 CH3CCH3 OH You do not have to consider stereochemistry. You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. Sn [F ChemDoodleⓇarrow_forwardIdentify two alkenes that react with HBr to form 1-bromo-1-methylcyclohexane without undergoing a carbocation rearrangement.arrow_forward
- Kelerences Keview topic Draw a structural formula for the major product of the reaction shown. Cl2 H20 CH;CH2CH;CH=CHz • Show product stereochemistry IF the reactant alkene has both carbons of the double bond within a ring. Do not show stereochemistry in other cases. • If the reaction produces a racemic mixture, just draw one stereoisomer. 2reg 2reg M) ots 2reg pts 2req 1 pts 2req 1 pts 2reg ...1 pts ChemDoodle Previous Next> Save and Exit APR tv MacBook Air 80 DII DD F3 F4 FS F6 F7 F8 F10 F1 F12 #3 $ & + 3 4 6. 7 8 %3D dele E Y | { } P [ F G H. J K C V N M ? .. .. * C0 Barrow_forwardWhich of the following reagents would you use to prepare Chlorocyclopentane from cyclopentene? OC2 / H20 NBS / H20, DMSO OCh/ CH,Cl2 HCIarrow_forwardFor each reaction, decide whether substitution or elimination (or both) is possible, andpredict the products you expect. Label the major products. chlorocyclohexane + NaOCH3 in CH3OHarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





