
(a)
Interpretation: The product formed from the ring-closing metathesis of given compound is to be drawn, and the synthesis of metathesis starting material using
Concept introduction: The ring-closing metathesis (RCM) by Grubbs catalyst occurs, when the starting material is diene. This reaction is facilitated under high-dilution condition as it favors intramolecular metathesis instead of intermolecular metathesis.
Answer to Problem 26.48P
The product formed from the ring-closing metathesis of given compound is,
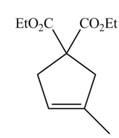
The synthesis of metathesis starting material using
Explanation of Solution
The given compound is a diene.
The ring-closing metathesis (RCM) by Grubbs catalyst occurs, when the starting material is diene. This reaction is facilitated under high-dilution condition as it favors intramolecular metathesis instead of intermolecular metathesis.
The product formed from the ring-closing metathesis of given compound is drawn in Figure 1.
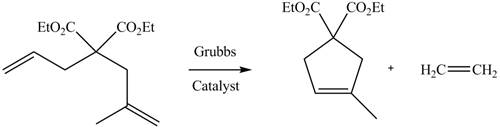
Figure 1
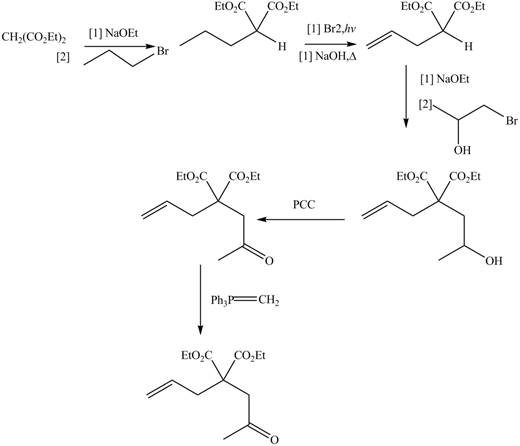
The synthesis of metathesis starting material involves five steps. The first step is reaction of
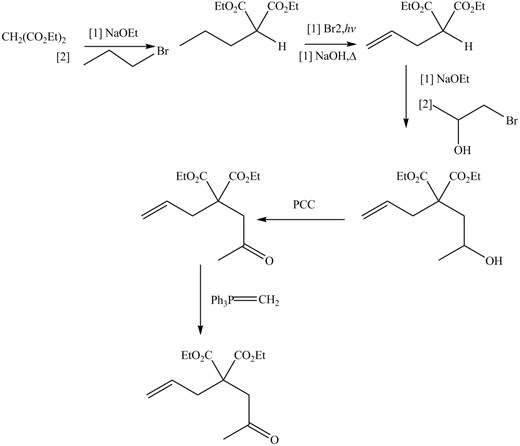
Figure 2
The product formed from the ring-closing metathesis of given compound is drawn in Figure 1. The synthesis of metathesis starting material using
(b)
Interpretation: The product formed from the ring-closing metathesis of given compound is to be drawn, and the synthesis of metathesis starting material using
Concept introduction: The ring-closing metathesis (RCM) by Grubbs catalyst occurs, when the starting material is diene. This reaction is facilitated under high-dilution condition as it favors intramolecular metathesis instead of intermolecular metathesis.
Answer to Problem 26.48P
The product formed from the ring-closing metathesis of given compound is,
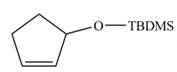
.The synthesis of metathesis starting material using
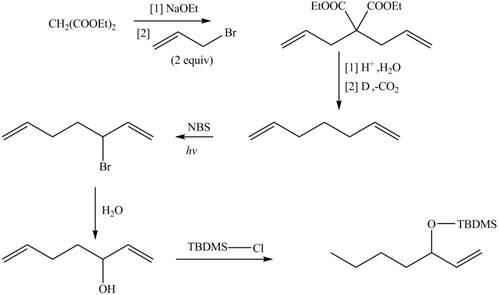
Explanation of Solution
The given compound is a diene.
The ring-closing metathesis (RCM) by Grubbs catalyst occurs, when the starting material is diene. This reaction is facilitated under high-dilution condition as it favors intramolecular metathesis instead of intermolecular metathesis.
The product formed from the ring-closing metathesis of given compound is drawn in Figure 3.
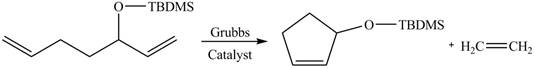
Figure 3
The synthesis of metathesis starting material involves five steps.The first step is reaction of
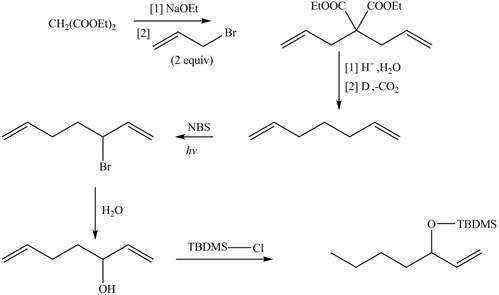
Figure 4
The product formed from the ring-closing metathesis of given compound is drawn in Figure 3. The synthesis of metathesis starting material using
Want to see more full solutions like this?
Chapter 26 Solutions
PKG ORGANIC CHEMISTRY
- Devise a synthesis of each compound from benzene. You may also use any organic compounds having four carbons or fewer, and any required inorganic reagents. Co,CH3 a. b.arrow_forwardDraw the product formed from the ring-closing metathesis of each compound. Then, devise a synthesis of each metathesis starting material using any of the following compounds: CH2(CO2Et)2, alcohols with four or fewer carbons, and any needed organic or inorganic reagents.arrow_forwardDraw the products formed from nitration of each compound. OH он b. а. CH3arrow_forward
- Devise a synthesis of each compound using CH3CH2CH=CH2 as the starting material. You may use any other organic compounds or inorganic reagents.arrow_forwardDraw compounds B and C from the following reaction scheme. excess CH3I Ag20, H20 heat Aarrow_forwardDevise a synthesis of CH,CH,C=CCH,CH,OH using CH,CH,CH-CH, as the starting material. You may use any other organiC compounds or inorganic reagents. Be sure to anSwer all parts. HO Step 1: HCL 2 equiv NOCI Intermediatel: draw structure Step 2: Intermediate 2: draw atructure Step 3: CH,CH;CH; Br NHy Intermediate 3: draw atructure .. Step 4: CHCH,CHarrow_forward
- Devise a synthesis of each compound. You may use HC=CH, ethylene oxide, and alkyl halides as organic starting materials and any inorganic reagents.arrow_forwardDevise a synthesis of each compound. You may use HC ≡ CH, ethylene oxide, and alkyl halides as organic starting materials and any inorganic reagents.arrow_forwardDevise a synthesis of each compound using CH3CH3 as the only source of carbon atoms. You may use any other required organic or inorganic reagents.arrow_forward
- Devise a synthesis of each compound from CH3CH2OH as the only organic starting material; that is, every carbon in the product must come from a molecule of ethanol. You may use any other needed inorganic reagents.arrow_forwardDevise a synthesis of each compound from benzene. You may use alcohols with one or two carbons and any inorganic reagents. a. qe Br H Br b. C. N OHarrow_forwardDraw the major organic product formed in the reaction. HBr CH=CH-CH=CH2 (1 equiv.)arrow_forward
