
a)
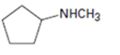
Interpretation:
The major product expected when the
Concept introduction:
Hofmann’s rule is applicable to elimination reactions involving positively charged and bulky leaving groups. According to this rule, when two possibilities exist in an elimination reaction, the hydrogen is eliminated from the carbon with more number of hydrogen atoms so that a less substituted
b)
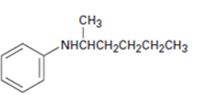
Interpretation:
The major product expected when the amine shown undergoes Hofmann elimination is to be given.
Concept introduction:
Hofmann’s rule is applicable to elimination reactions involving positively charged and bulky leaving groups. According to this rule, when two possibilities exist in an elimination reaction, the hydrogen is eliminated from the carbon with more number of hydrogen atoms so that a less substituted alkene will be the major product.
c)
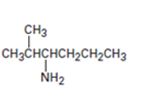
Interpretation:
The major product expected when the amine shown undergoes Hofmann elimination is to be given.
Concept introduction:
Hofmann’s rule is applicable to elimination reactions involving positively charged and bulky leaving groups. According to this rule, when two possibilities exist in an elimination reaction, the hydrogen is eliminated from the carbon with more number of hydrogen atoms so that a less substituted alkene will be the major product.
Trending nowThis is a popular solution!

Chapter 24 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- Provide the steps to convert the following aldehyde to the given amine product.arrow_forwardNH₂ The following reaction will give 3 different organic products. Sketch the three products. CH3Br Provide a synthesis for the following amine using a Gabriel synthesis. NH₂arrow_forwardDraw the structures of the carbonyl compound and the amine that will react at pH 5 to yield the product shown below: pH 5 carbonyl compound aminearrow_forward
- Hydrolysis of amides is an important reaction, because it represents the first step in the digestion of dietary protein. Which of the following compounds is formed as a result of the hydrolysis of N,N-diethylpropanamide as shown below? CH3CH2 N-CH2CH3 + H₂O CH2CH3 HCI OA) B) CH3CH2 NH2 HO N-CH2CH3 CH2CH3 H CH3CH2 NHCH2CH3 D)arrow_forwardShow how Gabriel syntheses are used to prepare the following amines. ) hexan-1-aminearrow_forwardOne of the first steps in the synthesis of Risperidone is the electrophilic aromatic substitution shown below. Draw the detailed reaction mechanism for this transformation:arrow_forward
- Atenolol is a beta blocker medication primarily used to treat high blood pressure and heart- associated chest pain. Other uses include the prevention of migraines and treatment of certain irregular heart beats It is taken by mouth or by injection into a vein. It can also be used with other blood pressure medications. OH .N. CH3 ČH3 H2N° suggest industrial synthetic approach towards synthesis of this drug (atenolol) and provide reasons and advantages of your proposed synthetic approach.arrow_forwardThe structure of an ester could either be A or B: CH3 O O CH3 T || -C-OCH3 CH3-C CH3-C-O-C-CH3 CH3 CH3 A B Its ¹H NMR spectrum consists of two peaks at 80.9 and 83.6 (relative areas 3:1). Which compound is it? Describe the spectrum that would be expected if it had been the other ester.arrow_forward9. What is the major Hofmann elimination product formed from the following amine? CH3 CHs- NH2 CH,CH, Č(CH3)3arrow_forward
- The compound below can form a hemiacetal by reacting with itself in solution. What is the structure of this hemiacetal? HO H. о но H о но Н НО ОН ОН ОНarrow_forwardWhat products will be obtained if the following compound is hydrolyzed completely in an aqueous solution of HCL?arrow_forwardDraw the major product formed from Hofmann elimination of the following amine.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

