
Visual Essentials of Anatomy &Physiology
1st Edition
ISBN: 9780321780775
Author: Martini, Frederic
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2.4, Problem 3SR
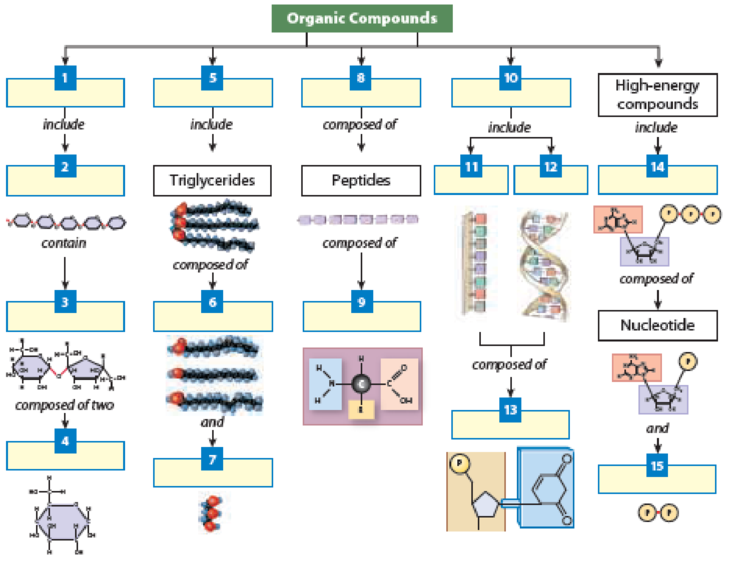
Concept map: Use each of the following terms once to fill in the blank boxes to correctly complete the map.
- lipids
- carbohydrates
nucleic acids - disaccharides
- RNA
- fatty acids
- phosphate groups
- glycerol
- polysaccharides
- proteins
- monosaccharides
- ATP
- amino acids
- DNA
nucleotides
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Describe the structures, functions, properties and types of lipids (e.g. fats, phospholipids, steroids). Recognize examples of the different types of lipids.
Describe functions of carbohydrates.
Describe the polymerization process of carbohydrates.
Describe the degradation process of carbohydrates.
Identify two (2) functions of lipids in the body. Explain each function in 2 sentences.
Chapter 2 Solutions
Visual Essentials of Anatomy &Physiology
Ch. 2.1 - Prob. 1LOCh. 2.1 - Explain the relationship between electrons and...Ch. 2.1 - Prob. 3LOCh. 2.1 - Prob. 1.1RCh. 2.1 - Prob. 1.2RCh. 2.1 - Prob. 1.3RCh. 2.1 - Prob. 2.1RCh. 2.1 - Explain why the atoms of inert elements do not...Ch. 2.1 - Prob. 2.3RCh. 2.1 - Prob. 3.1R
Ch. 2.1 - Prob. 3.2RCh. 2.1 - Prob. 3.3RCh. 2.1 - Fill-in: Fill in the missing information in the...Ch. 2.1 - Indicate which of the following molecules are also...Ch. 2.1 - Indicate which of the following molecules are also...Ch. 2.1 - Indicate which of the following molecules are also...Ch. 2.1 - Prob. 2.4SRCh. 2.1 - Matching: Match each lettered term with the most...Ch. 2.1 - b ____________________ Located in the nucleus,...Ch. 2.1 - c ____________________ Atoms of two more different...Ch. 2.1 - Matching: Match each lettered term with the most...Ch. 2.1 - Matching: Match each lettered term with the most...Ch. 2.1 - f ____________________ Type of chemical bond...Ch. 2.1 - Matching: Match each lettered term with the most...Ch. 2.1 - Matching: Match each lettered term with the most...Ch. 2.1 - Matching: Match each lettered term with the most...Ch. 2.1 - Matching: Match each lettered term with the most...Ch. 2.1 - Prob. 3.11SRCh. 2.1 - Prob. 3.12SRCh. 2.1 - Section integration: Describe how the following...Ch. 2.1 - Section integration: Describe how the following...Ch. 2.1 - Section integration: Describe how the following...Ch. 2.2 - Use chemical notation to symbolize chemical...Ch. 2.2 - Prob. 2LOCh. 2.2 - Prob. 1.1RCh. 2.2 - Prob. 1.2RCh. 2.2 - Prob. 1.3RCh. 2.2 - Prob. 2.1RCh. 2.2 - Prob. 2.2RCh. 2.2 - C. What is the source of energy that converts...Ch. 2.2 - Prob. 1.1SRCh. 2.2 - Prob. 1.2SRCh. 2.2 - Prob. 1.3SRCh. 2.2 - Prob. 1.4SRCh. 2.2 - Prob. 2SRCh. 2.2 - Prob. 3.1SRCh. 2.2 - Prob. 3.2SRCh. 2.2 - Prob. 4.1SRCh. 2.2 - Prob. 4.2SRCh. 2.2 - Prob. 4.3SRCh. 2.2 - Prob. 4.4SRCh. 2.2 - Prob. 4.5SRCh. 2.2 - Prob. 4.6SRCh. 2.2 - Prob. 4.7SRCh. 2.2 - Prob. 4.8SRCh. 2.2 - Prob. 5SRCh. 2.3 - Prob. 1LOCh. 2.3 - Prob. 2LOCh. 2.3 - Prob. 1.1RCh. 2.3 - Prob. 1.2RCh. 2.3 - Prob. 1.3RCh. 2.3 - Prob. 2.1RCh. 2.3 - Prob. 2.2RCh. 2.3 - Prob. 2.3RCh. 2.3 - Prob. 1.1SRCh. 2.3 - Prob. 1.2SRCh. 2.3 - Prob. 1.3SRCh. 2.3 - Prob. 1.4SRCh. 2.3 - Prob. 2.1SRCh. 2.3 - Prob. 2.2SRCh. 2.3 - Prob. 2.3SRCh. 2.3 - Prob. 2.4SRCh. 2.3 - Prob. 2.5SRCh. 2.3 - Prob. 2.6SRCh. 2.3 - Prob. 2.7SRCh. 2.3 - Prob. 2.8SRCh. 2.3 - Prob. 2.9SRCh. 2.3 - Prob. 2.10SRCh. 2.3 - Prob. 3.1SRCh. 2.3 - Prob. 3.2SRCh. 2.3 - Prob. 3.3SRCh. 2.3 - Prob. 3.4SRCh. 2.3 - Prob. 3.5SRCh. 2.3 - Prob. 4SRCh. 2.4 - Discuss the structures and functions of...Ch. 2.4 - Prob. 2LOCh. 2.4 - Prob. 3LOCh. 2.4 - Discuss protein structure and the essential...Ch. 2.4 - Prob. 5LOCh. 2.4 - Prob. 6LOCh. 2.4 - Prob. 7LOCh. 2.4 - Prob. 1.1RCh. 2.4 - Prob. 1.2RCh. 2.4 - Prob. 1.3RCh. 2.4 - Prob. 2.1RCh. 2.4 - Prob. 2.2RCh. 2.4 - Prob. 2.3RCh. 2.4 - Prob. 3.1RCh. 2.4 - Prob. 3.2RCh. 2.4 - Prob. 3.3RCh. 2.4 - Prob. 4.1RCh. 2.4 - Prob. 4.2RCh. 2.4 - Prob. 4.3RCh. 2.4 - Prob. 5.1RCh. 2.4 - Prob. 5.2RCh. 2.4 - Prob. 5.3RCh. 2.4 - Prob. 6.1RCh. 2.4 - Prob. 6.2RCh. 2.4 - Prob. 6.3RCh. 2.4 - Prob. 7.1RCh. 2.4 - Prob. 7.2RCh. 2.4 - Prob. 7.3RCh. 2.4 - Prob. 1.1SRCh. 2.4 - Prob. 1.2SRCh. 2.4 - Prob. 1.3SRCh. 2.4 - Prob. 1.4SRCh. 2.4 - Prob. 1.5SRCh. 2.4 - Prob. 1.6SRCh. 2.4 - Prob. 1.7SRCh. 2.4 - Prob. 1.8SRCh. 2.4 - Prob. 1.9SRCh. 2.4 - Prob. 1.10SRCh. 2.4 - Prob. 1.11SRCh. 2.4 - Prob. 2.1SRCh. 2.4 - Prob. 2.2SRCh. 2.4 - Prob. 2.3SRCh. 2.4 - Prob. 2.4SRCh. 2.4 - Concept map: Use each of the following terms once...Ch. 2 - Prob. 1CRQCh. 2 - An important buffer system in the human body...Ch. 2 - Prob. 3CRQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Arrange each ingredient accordingly. [Ingredients: pumpkin, romaine lettuce, tomato, ricotta cheese, white vinegar, pecans] Monosaccharides (sugars) Polysaccharides (starch and/or cellulose) Lipids (indicate if each is saturated, unsaturated, trans) Proteins Nucleic Acidsarrow_forwardMatching: Basic Chemistry/Biochemistry Terms Terms proteins anion carbohydrates cation organic glucose ion nucleic acids lipids metabolism waxes, oils, fats, and phospholipids Is always positively charged Is always negatively charged Double layer component of the cell membrane Hydrophobic and do not dissolve in water Assembled by ribosomes according to DNA's instructions Carbohydrate needed for the production of ATP within a cell Used to move large or charged molecules through the cell membrane Term that includes all molecules of living things identifying them as containing carbon Molecule used as a quick release of energy; needed for cellular respirationarrow_forwardProvide the smallest molecular unit of most lipids, proteins, and nucleic acids. Keep in mind that lipids are a little tricky.arrow_forward
- Describe the structures, functions and properties of carbohydrates (including monosaccharides, disaccharides, and polysaccharides). Give specific examples of biologically important carbohydrates.arrow_forwardMatch the given descriptions to the type of lipids.arrow_forwardDraw a lipid structure. Properly label the polar and non-polar ends of the representation of a lipid below:arrow_forward
- A. Fill in the table by comparing the following biological molecules. Biological Molecules Basic Biological Drawing/Illustration Unit Importance of monomer Carbohydrates Proteins Lipids Nucleic Acidsarrow_forwardBriefly describe 5 major types of lipids.arrow_forward1. What elements do lipids contain? 2. Identify two (2) functions of lipids in the body. Explain each function in 2 sentences. 3. Label the parts of the below lipid molecule. Is this a saturated or unsaturated lipid? I-0- HIC I-0)-I C-C- I ннн H-C-H C-C H-C-I H-C-H I-C-I HH C-C-CH I H с 4 Harrow_forward
- 26. Complete the table below to summarize your understanding of macromolecules. CHEMICAL NAME OF POLYMER NAME OF ELEMENTS FUNCTIONS MONOMER(S) IT'S MADE OF monosaccharides carbohydrate starch polysaccharides glycogen cellulose oils/fats lipids phospholipids steroids proteins DNA nucleic acids MRNA ATP 11arrow_forwardState six roles of lipids in the body, and give an exampleof each.arrow_forwardConcerning carbohydrates, it is INCORRECT to affirm that: Monosaccharides (simple sugars that contain from 3 to 7 carbon atoms) Disaccharides (simple sugars formed from the combination of two monosaccharides by dehydration synthesis). Polysaccharides (from tens to hundreds of monosaccharides joined by dehydration synthesis). Examples of Polysaccharides are Sucrose (table sugar) = glucose + fructose. Lactose (milk sugar) = glucose + galactose. Maltose = glucose + glucosearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
Macromolecules | Classes and Functions; Author: 2 Minute Classroom;https://www.youtube.com/watch?v=V5hhrDFo8Vk;License: Standard youtube license