
Student Study Guide and Solutions Manual for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
8th Edition
ISBN: 9781305864504
Author: Brent L. Iverson, Sheila Iverson
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 23.5, Problem FQ
Interpretation Introduction
Interpretation:
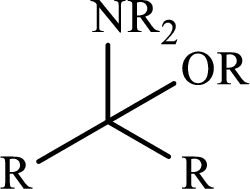
Name of the organic
Concept introduction:
An
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
KINDLY HELP ME WITH THIS PROBLEM MULTIPLE CHOICE THANK YOU !
How can the structure of the following molecule be changed in order to make it more acidic?
A. Replacing the CI substituent with OH
8. Moving the CI substituent to the C position
C. Moving the CI substituent to the C position
D. Replacing the CI substituent with an NH₂
явон
The most common reaction involving the carboxylic acids and its derivatives is related to the presence of an acyl group.
In the structure shown below, which group corresponds to the alkyl group of the acyl structure?
A. n-propyl group
8. Chloro group
C. Isobutyl group
D. Carbonyl group
An aldehyde commonly exhibits a nucleophilic addition type of reaction. When a nucleophile attacks a carbonyl carbon,
what happens to the oxygen atom in the structure? Refer to the structure below.
A. Oxygen atom obtains a net negative charge.
B. Oxygen atom becomes more electronegative.
C. Oxygen atom acts as the new electrophile.
D. Oxygen atom transforms to an alkoxide group.
1. There is one compound (that contains only one carbonyl group) that gives both a positive iodoform test and a positive Tollens’ test. This compound is not included in the table of unknowns. Use your knowledge of the test to predict this compound and draw it. Hint: the molecular weight of the compound is less than 60
2. Draw the structure of a molecule, an unknown aldehyde or ketone listed in the tables, that would show a negative Tollens’ test and a negative iodoform test. There is more than one correct answer. Draw one!
3. You acquired the following data: bp 173 °C; – Tollens’, + iodoform tests; semicarbazone mp 105 °C. You are very confident that your unknown is 2- octanone. Data is spot on, excluding the semicarbazone mp. You suspect the suppressed mp is due to wet crystals. What process or technique (other than continued oven drying) can help you obtain more pure semicarbazone and thus a more accurate mp for comparison? Please write answer in box at right.
Chapter 23 Solutions
Student Study Guide and Solutions Manual for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
Ch. 23.1 - Prob. 23.1PCh. 23.2 - Prob. 23.2PCh. 23.2 - Prob. 23.3PCh. 23.2 - Prob. 23.4PCh. 23.5 - Prob. 23.5PCh. 23.5 - Prob. AQCh. 23.5 - What is the hybridization of the nitrogen in...Ch. 23.5 - Prob. CQCh. 23.5 - The pKas of the conjugate acids of aniline and...Ch. 23.5 - Prob. EQ
Ch. 23.5 - Prob. FQCh. 23.5 - Prob. GQCh. 23.5 - Select the stronger acid from each pair of...Ch. 23.6 - Prob. 23.7PCh. 23.6 - Prob. 23.8PCh. 23.6 - Prob. 23.9PCh. 23.7 - Prob. 23.10PCh. 23.8 - Prob. 23.11PCh. 23.8 - Prob. 23.12PCh. 23.8 - Prob. 23.13PCh. 23.9 - Prob. 23.14PCh. 23.10 - In Example 23.15, you considered the product of...Ch. 23 - Prob. 23.16PCh. 23 - Prob. 23.17PCh. 23 - Prob. 23.18PCh. 23 - Prob. 23.19PCh. 23 - Prob. 23.20PCh. 23 - Prob. 23.21PCh. 23 - Prob. 23.22PCh. 23 - Account for the formation of the base peaks in...Ch. 23 - Prob. 23.24PCh. 23 - Select the stronger base from each pair of...Ch. 23 - The pKa, of the conjugate acid of morpholine is...Ch. 23 - Which of the two nitrogens in pyridoxamine (a form...Ch. 23 - Prob. 23.28PCh. 23 - Prob. 23.29PCh. 23 - Prob. 23.30PCh. 23 - Prob. 23.31PCh. 23 - Suppose you have a mixture of these three...Ch. 23 - Prob. 23.33PCh. 23 - Prob. 23.34PCh. 23 - Prob. 23.35PCh. 23 - Prob. 23.36PCh. 23 - Prob. 23.37PCh. 23 - (S)-Glutamic acid is one of the 20 amino acid...Ch. 23 - Prob. 23.39PCh. 23 - Propose a structural formula for the compound...Ch. 23 - Prob. 23.41PCh. 23 - The pyrolysis of acetic esters to give an alkene...Ch. 23 - Propose steps for the following conversions using...Ch. 23 - Show how to bring about each step in this...Ch. 23 - Show how to bring about each step in the following...Ch. 23 - Prob. 23.48PCh. 23 - Prob. 23.49PCh. 23 - Methylparaben is used as a preservative in foods,...Ch. 23 - Prob. 23.51PCh. 23 - Prob. 23.52PCh. 23 - Propose a synthesis for the systemic agricultural...Ch. 23 - Prob. 23.54PCh. 23 - Several diamines are building blocks for the...Ch. 23 - Prob. 23.56PCh. 23 - Prob. 23.57PCh. 23 - Prob. 23.58PCh. 23 - Prob. 23.59PCh. 23 - Following is a retrosynthesis for the coronary...Ch. 23 - Prob. 23.61PCh. 23 - Prob. 23.62PCh. 23 - Given this retrosynthetic analysis, propose a...Ch. 23 - Prob. 23.64PCh. 23 - Following is a series of anorexics (appetite...Ch. 23 - Prob. 23.66PCh. 23 - Prob. 23.67PCh. 23 - Show how the synthetic scheme developed in Problem...Ch. 23 - Prob. 23.69PCh. 23 - Prob. 23.70PCh. 23 - Prob. 23.71PCh. 23 - Prob. 23.72P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Complete each acid-base reaction and predict whether the position of equilibrium lies toward the left or toward the right. (a) CH3CCH+CH3CH2ONa+CH3CH3OH (b) CH3CCCH2CH2OH+Na+NH2NH3(l)arrow_forwardI need the answer as soon as possiblearrow_forwardb. Which of the following is the structure of acetic anhydride? si OH c. When 4-ethoxyaniline reacts with acetic anhydride, what new functional group or type of compound is formed? O acid anhydride acetal amide O esterarrow_forward
- Draw the three possible resonance structures for an acid bromide, RCOBr. Then decide if RCOBr is more or less stabilized by resonance than a carboxylic acid (RCOOH).arrow_forwardFirst, draw your chosen compounds including any non-bonding electrons and non-zero formal charges Circle and name any pH-sensitive functional groups. Choose an organic solvent that will dissolve both or your compounds and an aqueous solution (i.e. conc. aqueous NaOH, 5% aq. NaHCO3, or 3M HCl) that would successfully separate the two compounds selected.arrow_forwardPlease show the structure of the missing substances in each of the following acid base equilibria.arrow_forward
- 1. Predict whether the compound below will test positive for each of the following qualitative tests: a) Tollens’ test, b) Benedict’s test 2. Provide at least one (1) test that could differentiate n-pentanol and pentanal and give the expected visible result/s. [n-pentanol and pentanal]arrow_forwardThe acetal shown below is composed of which of the following original compounds?arrow_forward1 1. Write the Balanced Chemical equation for each of the reaction step in the isolation of your org nic base compound. Use the structure of the specific compound you identified as organic base. Don't use the molecular formula. Indicate their physical state. cid-Cinnamic Acid Base-5-chloro-2-methoxynaline Neutral-P-dibromobenzene Iarrow_forward
- Determine whether the following reaction is an example of a nucleophilic substitution reaction: 8+ + H2O Molecule A Molecule B OH ++H Is this a nucleophilic substitution reaction? If this is a nucleophilic substitution reaction, answer the remaining questions in this table. What word or two-word phrase is used to describe the role Molecule A plays in this reaction? ☐ What word or two-word phrase is used to describe the role Molecule B plays in this reaction? What word or two-word phrase is used to describe the role of the carbon labeled with the 6+ symbol? What word or two-word phrase is used to describe the role of the atom or group highlighted in red? ☐ ☐ ☐ Yes Noarrow_forwardDo not give handwriting solution.arrow_forwardWhat is the correct IUPAC name for this compound? Select an answer and submit. For keyboard navigation, use the up/down arrow keys to select an answer. 7-bromo-4-methylheptanoic acid b 6-bromo-3-methylhexanoic acid C 1-bromo-4-methylheptanoic acid d 6-bromo-3-methylhexoicacid Br CH3 COOHarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning