
Concept explainers
Which compound in each pair is the stronger acid?
a.
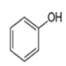 or
or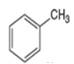
Trending nowThis is a popular solution!

Chapter 2 Solutions
Organic Chemistry (6th Edition)
- In each pair, select the stronger acid. (a) Pyruvic acid (pKa 2.49) or lactic acid (pKa 3.08) (b) Citric acid (pKa1 3.08) or phosphoric acid (pKa1 2.10)arrow_forwardWhich is a stronger acid? a) HCl or HBr b) CH3CH2CH2N+H3 or CH3CH2CH2O+H2 d)CH3CH2CH2OH or CH3CH2CH2SHarrow_forwardWhich is a stronger base? a. CH3O− or CH3S− b. H2O or HO− c. H2O or NH3 d. CH3CO− or CH3O−arrow_forward
- Which is the stronger base? a. Br- or I- b. CH3O- or CH3S-c. CH3CH2O− or CH3COO−d. H2C =CH e. FCH2CH2COO− or BrCH2CH2COO− f. ClCH2CH2O− or Cl2CHCH2O−arrow_forwardWhich is the more stable base? a. Br− or I− b. CH3O− or CH3S− c. CH3CH2O− or CH3COO− d. H2C CH or HC C− e. FCH2CH2COO− or CH2CH2COO- f. CLCH2CH2O- or CL2CHCH2O-arrow_forwardcompared to HCO3-, H2co3 has a A Stronger conjugated base B Weaker conjugated base C Higher ka D Lower ka E Both b and carrow_forward
- Hypochlorous acid reacts with sodium bicarbonate (also known as sodium hydrogen carbonate). What are the products of this reaction? (A) NaCIO4 + H2CO2 (B) NaCIO4 + H2CO3 (C) NaCIO + H2CO2 (D) NaCIO + H2CO3arrow_forwardGiven the following acid-base equilibria:HCOOH + H2O ---> H3O+ + HCOO-CH3NH3+ + H2O---> H3O+ + CH3NH2HSO4- + H2O---> H3O+ + SO42-Circle strongest acid and cross out the weakest.HCOOH HSO4- CH3NH3+Circle strongest base and cross out the weakest.HCOO- SO2- CH3NH2arrow_forwardwhich compound is the stronger acid a. CH3CONH2 or CH3COOH b. 1-hydroxy-3methylbenzene or 1-hydroxyl-3-trifluoromethylbenzene c. methanol or phenol d. acetic acid vs oxalic acidarrow_forward
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


