
Organic Chemistry
7th Edition
ISBN: 9780321803221
Author: Paula Y. Bruice
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 23, Problem 36P
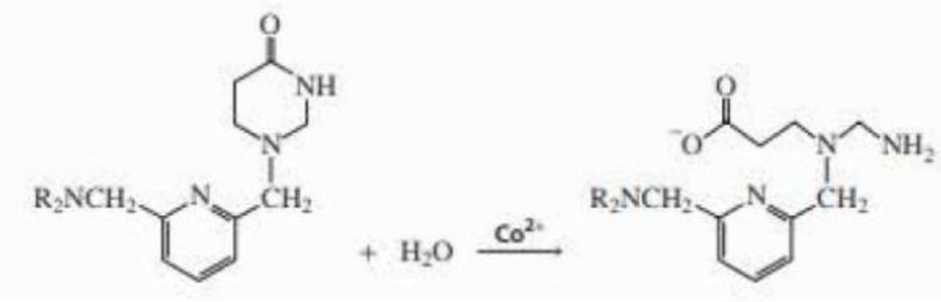
Co2+ catalyzes the hydrolysis of the lactam shown here. Propose a mechanism for the metal-ion catalyzed reaction.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Draw the mechanism for acid-catalyzed ester hydrolysis.
Co2+ catalyzes the hydrolysis of the lactam shown here. Propose a mechanism for the metal-ion catalyzed reaction.
The following three derivatives of succinimide are anticonvulsants and have found use in the treatment of epilepsy, particularly petit mal seizures.
Ph
Ph
`N'
`N'
ČH3
ČH3
Methsuximide
Ethosuximide
Phensuximide
Following is a synthesis of phensuximide.
CN
Ph
CN
Ph
CN
1. NaOH, H2O
2. HC, Н20
NaOEt
KCN
Ph-CHO
cOOEt
H
cOOEt
NC
COOEt
3. Нeat
Ethyl
cyanoacetate
(A)
(B)
Benzaldehyde
Ph
Ph
Ph
CH;NH2
НООС
СООН
Et0oC
COOEt
`N'
(C)
(D)
ČH3
Phensuximide
Methsuximide is formed by a similar pathway to that shown for phensuximide. Draw the structure of the compound that reacts with ethyl cyanoacetate in the synthesis of
methsuximide.
Chapter 23 Solutions
Organic Chemistry
Ch. 23.2 - Compare each of the mechanisms listed here with...Ch. 23.2 - Prob. 3PCh. 23.2 - Prob. 4PCh. 23.3 - a. Draw the mechanism for the following reaction...Ch. 23.5 - Prob. 7PCh. 23.5 - Propose a mechanism for the Co2+ catalyzed...Ch. 23.6 - Prob. 9PCh. 23.7 - Prob. 10PCh. 23.7 - Prob. 12PCh. 23.7 - Prob. 13P
Ch. 23.9 - Which of the following amino acid side chains can...Ch. 23.9 - Which of the following C-terminal peptide bonds is...Ch. 23.9 - Carboxypeptidase A has esterase activity as well...Ch. 23.9 - Arginine and lysine side chains fit into trypsins...Ch. 23.9 - Explain why serine proteases do not catalyze...Ch. 23.10 - If H2 18O is used in the hydrolysis reaction...Ch. 23.10 - When apples that have been cut are exposed to...Ch. 23.11 - Prob. 22PCh. 23.11 - The pHactivity profile for glucose-6-phosphate...Ch. 23.11 - Draw the pH-activity profile for an enzyme that...Ch. 23.12 - Prob. 25PCh. 23.12 - Draw the mechanism for the hydroxide ion-catalyzed...Ch. 23.12 - What advantage does the enzyme gain by forming an...Ch. 23.12 - Prob. 28PCh. 23.12 - Aldolase shows no activity if it is incubated with...Ch. 23 - Which of the following parameters would be...Ch. 23 - Prob. 30PCh. 23 - Prob. 31PCh. 23 - Prob. 32PCh. 23 - Indicate the type of catalysis that is occurring...Ch. 23 - The deuterium kinetic isotope effect (KH2O/KD2O)...Ch. 23 - Prob. 35PCh. 23 - Co2+ catalyzes the hydrolysis of the lactam shown...Ch. 23 - there are two kinds of aldolases. Class I...Ch. 23 - Prob. 38PCh. 23 - The hydrolysis of the ester shown here is...Ch. 23 - Prob. 40PCh. 23 - At pH = 12, the rate of hydrolysis of ester A is...Ch. 23 - 2-Acetoxycyclohexyl tosylate reacts with acetate...Ch. 23 - Proof that an imine was formed between aldolase...Ch. 23 - Prob. 44PCh. 23 - a. Explain why the alkyl halide shown here reacts...Ch. 23 - Triosephosphate isomerase (TIM) catalyzes the...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Ethanolamine ammonia lyase, a coenzyme B12–requiring enzyme, catalyzes the following reaction. Propose a mechanism for this reaction.arrow_forwardNaturally occurring compounds called cyanogenic glycosides, such as lotaustralin, release hydrogen cyanide, HCN, when treated with aqueous acid. The reaction occurs by hydrolysis of the acetal linkage to form a cyanohydrin, which then expels HCN and gives a carbonyl compound. (a) Show the mechanism of the acetal hydrolysis and the structure of the cyanohydrin that results. (b) Propose a mechanism for the loss of HCN, and show the structure of the carbonyl compound that forms.arrow_forwardWhen cyclohexanone is heated in the presence of a large amount of acetone cyanohydrin and a small amount of base, cyclohexanone cyanohydrin and acetone are formed. Propose a mechanism.arrow_forward
- The addition of a nucleophilic group like a thiol (-SH), or an unprotonated amino group (-NH₂) to an a,ß-unsaturated ketone is important for understanding how NAPQI reacts with glutathione, N-acetylcysteine, or protein amino acid side chains containing -SH or -NH2 groups. In this problem we will add mercaptoethanol to p-benzoquinone. p-Benzoquinone will be a simple model of NAPQI. +H H a single uncharged addition product This reaction will proceed in steps. First the H-S-R will add to the unsaturated ketone to give a charged addition product. Next, a simple proton transfer will form an uncharged adduct in which the 6-membered ring contains one carbonyl and a second enolic hydroxyl. The keto form next will tautomerize to its more stable enol form, and in doing so the 6-membered ring becomes aromatic with 6-π electrons. See page 14 of the lecture notes for Lecture 9 How ADME affects preclinical Drug Design February 24, 2023, where I have shown the mechanism for this addition and give the…arrow_forwardWhen an α-hydroxy amide is treated with Br2 in aqueous NaOH under Hofmann rearrangement conditions, loss of CO2 occurs and a chain-shortened aldehyde is formed. The mechanism involves the following steps: Base abstracts an acidic amide proton, yielding amide anion 1; The amide anion reacts with bromine in an α-substitution reaction to give N-bromoamide 2. Abstraction of the remaining amide proton by base gives a resonance-stabilized bromoamide anion 3; Rearrangement occurs to yield isocyanate 4; Water adds to the isocyanate to yield carbamic acid 5; Elimination of CO2 yields carbinolamine 6; Following proton transfer, expulsion of ammonia yields the final product aldehyde. Write out the mechanism and then draw the structures of bromoamide anion 3 and amide anion 1.arrow_forwardThe mechanism for acidic hydrolysis of a nitrile resembles the basic hydrolysis, exceptthat the nitrile is first protonated, activating it toward attack by a weak nucleophile (water).Under acidic conditions, the proton transfer (tautomerism) involves protonation on nitrogen followed by deprotonation on oxygen. Propose a mechanism for the acid-catalyzedhydrolysis of benzonitrile to benzamide.arrow_forward
- a OH CH2CNHCH3 T CH3 d OH CHCHNHCH3 CH3 Ephedrine can be synthesized via reductive amination plus other necessary reactions. Predict the necessary reactants and reagents that will give the intermediate and final products shown.arrow_forwardSelect the appropriate reagent and TWO possible substrates for the following reductive amination.arrow_forwardThe reagent diisobutylaluminum hydride (DIBALH) reduces esters to aldehydes. When nitriles are treated with DIBALH followed by mild acid hydrolysis, the product is also an aldehyde. Propose a mechanism for this reduction.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Enzymes - Effect of cofactors on enzyme; Author: Tutorials Point (India) Ltd;https://www.youtube.com/watch?v=AkAbIwxyUs4;License: Standard YouTube License, CC-BY
Enzyme Catalysis Part-I; Author: NPTEL-NOC IITM;https://www.youtube.com/watch?v=aZE740JWZuQ;License: Standard Youtube License